Tristan Loscha
Member
- Joined
- Dec 18, 2018
- Messages
- 2,206
Finally Confirmed! Vitamin D Nearly Abolishes ICU Risk in COVID-19
September 3, 2020
The first randomized controlled trial (RCT) of vitamin D in COVID-19 has just been published. The results are astounding: vitamin D nearly abolished the odds of requiring treatment in ICU. Although the number of deaths was too small to say for sure, vitamin D may actually abolish the risk of death from COVID-19.
The First Randomized Controlled Trial on Vitamin D and COVID-19
The trial was conducted at the Reina Sofía University Hospital in Córdoba, Spain. The trial included 76 patients with COVID-19 pneumonia. Although this is no longer the standard of care, all patients were treated with hydroxychloroquine and azithromycin and, when needed, a broad-spectrum antibiotic. Admission to the ICU was determined by a multidisciplinary committee consisting of intensive care specialists, pulmonologists, internal medicine specialists, and members of the ethics committee.
The patients were randomly allocated to receive or not receive vitamin D in a 2:1 ratio. This resulted in 50 patients in the vitamin D group and 26 patients in the control group.
The Vitamin D Treatment Protocol
The vitamin D was provided as oral calcifediol, also known as calcidiol, 25(OH)D, and 25-hydroxyvitamin D. This is a metabolite of vitamin D that our livers make. It is is the principle form of vitamin D that circulates in the blood, and we use it as a measure of vitamin D status.
Traces of 25(OH)D occur in food, and it is five times as potent as vitamin D. As described on page 255 of the 1997 DRI Report, 25(OH)D is given an international unit (IU) value that equates it to vitamin D. Whereas one microgram (mcg) of vitamin D is 40 IU, 1 mcg of 25(OH)D is 200 IU.
The treatment in this RCT was soft capsules of 532 mcg 25(OH)D on day 1 of admission to the hospital, followed by 266 mcg on days 3 and 7, and then 266 mcg once a week until discharge, ICU admission, or death.
This is equivalent to 106,400 IU vitamin D on day 1, 53,200 IU on days 3 and 7, and 53,200 IU weekly thereafter. If this were given as daily doses, it would be the equivalent of 30,400 per day for the first week, followed by a maintenance dose of 7,600 IU per day.
The vitamin D status of the patients was not measured. However, the average vitamin D status in this region of Spain during the time of year in which the study was conducted is 16 ng/mL. A single dose of 100,000 IU vitamin D tends to raise a 25(OH)D of 10 ng/mL into the 20-30 ng/mL range. My suspicion is that the bolus dosing in the first week brought the patients' vitamin D status into the 30-40 ng/mL range by the end of the week, and that most of the healing took place in the circa 40 ng/mL range.
The Results: Near Abolition of ICU Risk
The results are absolutely stunning. 50% of the control group (13 people) required admission to the ICU. Only 2% of those in the vitamin D group (one person) required admission to the ICU.
Expressed as relative risk, vitamin D reduced the risk of ICU admission 25-fold. Put another way, it eliminated 96% of the risk of ICU admission. Expressed as an odds ratio, which is a less intuitive concept but is often used in statistics because it gives an estimate of the effect of the treatment that would be constant across scenarios with different levels of risk, vitamin D reduced the odds of ICU admission by 98%. Either way, vitamin D practically abolished the need for ICU admission.
This was statistically significant at p<0.001, and the 95% confidence interval was 0.002-0.17. This means that the probability of observing differences this large or greater if there is no true effect of vitamin D is less than one in a thousand, and that the probability is 95% that the true effect lies somewhere between an 83% and a 99.8% reduction in the odds of ICU admission.
No matter how you slice it, the effect of vitamin D is extremely compelling.
Because the study is small, the potential confounding variables were not perfectly evenly distributed between the two groups. There was more high blood pressure in the control group, and there was a borderline greater number of patients with diabetes in the control group. Though not statistically significant, the vitamin D group had more people over the age of 60 and five times as many people with organ transplants or who were otherwise on immunosuppressive drugs. To account for all of these differences, they adjusted for them statistically. In the adjusted model, vitamin D still reduced the odds of ICU admission by 97%, with the 95% confidence interval ranging from a 75% to a 99.7% reduction in the odds.
Did Vitamin D Also Abolish the Risk of Death?
All of the vitamin D patients were discharged without complications. Half the control group was discharged without ICU admission. Among the other half, 11 were eventually discharged from ICU, and 2 died.
Although there were, thankfully, too few deaths to run statistics on, since patients generally would be admitted to the ICU before dying, and since vitamin D nearly abolished the risk of ICU admission, we can infer that in a larger study with more deaths, vitamin D would probably abolish or nearly abolish the risk of death.
Comparison With Observational Studies
These results are consistent with the first observational study on vitamin D, which found that 96% of severe and critical cases occurred at 25(OH)D under 30 ng/mL, whereas 97.5% of mild cases had 25(OH)D above 30 ng/mL.
This study couldn't measure the effect on mortality, but all two deaths were in the control group and the near abolition of ICU risk suggests that death would also be nearly abolished. This is consistent with the second observational study finding that only 4% of those with vitamin D status above 30 ng/mL died, while 88% of those with vitamin D status at 20-30 ng/mL died, and 99% of those with vitamin D status lower than that died.
It is less consistent with the weaker observational studies that came out later. For example, the fourth observational study found that prevalence of 25(OH)D below 20 ng/mL differed across three categories of severity judged by CT scan in males but not females. It is also less consistent with more recent studies in Iran and England. In Iran, 25(OH)D below 30 ng/mL only increased risk of severe infection by 21%, although mortality was roughly cut in half at that level. In England, ICU patients were half as likely to have 25(OH)D above 20 ng/mL as non-ICU patients, and mortality wasn't associated with vitamin D status.
In other words, the first RCT shows the effect of vitamin D is closer to the more extreme estimates of the first observational studies than it is to the more moderate estimates of the more recent studies.
The observational studies on infection risk are weaker than those on severity and mortality (see here, here, here, and here), but this RCT didn't look at infection risk.
The RCT contradicts the findings of the two Mendelian randomization studies. These found either no evidence for causality in the vitamin D/COVID-19 connection, or only weak evidence. However, these studies infer causality by looking at the effect of genetics. Genetics only explain 4.2% of the variation in 25(OH)D and many of the genes involved have non-specific, indirect relationships to vitamin D status. This RCT overwhelmingly takes precedence over the findings of the Mendelian randomization studies.
This Study Is the Single Most Important Vitamin D and COVID-19 Study
Since the first vitamin D study was released as a preprint* on April 23, we have been waiting for data that could settle whether the association between vitamin D and COVID-19 incidence, severity, and mortality is a causal one.
This study settles the question: yes, it is causal.
It is important for scientists to replicate each other's findings. Future studies with more diverse populations may reveal differences in the response between different populations. Future larger studies will more precisely refine the exact effect of vitamin D. Future studies with different dosing protocols, in different contexts (for example, without the use of hydroxychloroquine and azithromycin, or with the administration of other drugs or nutrients) will add nuances to our understanding of the effect of vitamin D.
However, given the degree to which it is nearly entirely harmless, and almost without exception beneficial, to maintain 25(OH)D above 30 ng/mL, it would be irresponsible not to interpret this study as definitive evidence of causality. At an absolute minimum, maintaining vitamin D status in this range should be part of the public health message to reduce COVID-19 risk, and 25(OH)D should be universally screened in all hospitals to be treated in anyone with COVID-19, and should be universally screened in all testing centers when antibodies and PCR testing is done, so that everyone knows not only their COVID-19 exposure but also their vitamin D status. If it's low, they should be given advice on how to bring it back up to normal.
How I've Changed My Position
On March 17, when I released Version 1 of The Food and Supplement Guide for the Coronavirus, I expressed concern that vitamin D might increase infection risk or severity by increasing ACE2, the entryway of SARS-CoV-2, the coronavirus that causes COVID-19, into our cells. In response to the first observational study of vitamin D released on April 23, which I covered in this newsletter on April 24, I released version 3 of the guide on April 28, in which I revised this stance and took the position that 25(OH)D should be maintained at 30-34 ng/mL.
As observational studies accumulated, they converged on the common point that 30-40 ng/mL is the sweet spot where infection risk, severity, and mortality are all lowest, without any risk of getting too much vitamin D.
I remained concerned that there might be some risk of a U-shaped curve, where risk increases at vitamin D levels higher than 40 ng/mL. Evidence for or against this was scant and contradictory. Data from Switzerland provided hints of an increased infection risk above 40 in those under 70 years old, but not in those older than 70. Data from Chicago suggested no difference in infection risk above 40 ng/mL, but it didn't separate the people by age. Data from Israel suggested that 25(OH)D above 53 ng/mL completely abolishes infection risk, but everyone with levels that high was under the age of 50. In the Israeli study, the relevant data were only in the young, and risk seemed abolished. In the Swiss study, the relevant data existed for all ages, and it suggested risk increased in the young. All of these studies concern infection risk, not severity or mortality, they all have too little data at high 25(OH)D levels, and they contradict each other.
While I remain agnostic whether there is some level of vitamin D above which infection risk is increased, the new RCT concerns severity and mortality. Although 25(OH)D levels weren't measured, they must have hit at least 40 ng/mL by the time the patients were released, and they possibly exceeded 50 ng/mL. That they gave these patients such massive doses of vitamin D without measuring their vitamin D levels, and that this nearly abolished the need for ICU admission, suggests that there should be no concern about a U-shaped curve with severity or mortality for short-term dosing of up to 8,000 IU per day over several weeks or for temporarily pushing 25(OH)D above 50 ng/mL during the course of treatment.
Given that observational studies around infection risk can consistently show that it is increased at low vitamin D status but cannot show any consistent picture at high vitamin D status, I am, for now, relieving myself of concern about this U-shaped curve. If high vitamin D status can almost eliminate severity and mortality, the off-chance that in some people at some high level it could increase infection risk should be tolerated.
You can see the evolution of my stance on vitamin D through these past posts:
I think the best way to take action on these findings is to maintain 25(OH)D in 30-40 ng/mL. This is the sweet spot according to the observational literature, and if we assume the patients in the Spanish RCT came in with 16 ng/mL, 30-40 ng/mL probably approximates their average blood level over the course of their recovery.
This is also consistent with other metrics of optimal vitamin D status. All-cause mortality is lowest at about 28 ng/mL according to one meta-analysis of eight European studies, seven conducted in the general population, pooling data from just under 27,000 people. Another widely circulated meta-analysis showing a bottoming-out in the 40-60 ng/mL range derived its data in that range entirely from a conference abstract. If that one outlier is removed, as can clearly be seen in Figure 4 of the paper, the bottoming out of all-cause mortality is below 40 ng/mL and probably close to 30 ng/mL. This is consistent with the amount needed to maximally suppress parathyroid hormone (PTH) in most people, which is the body's own sign of inadequacy in the vitamin D and calcium economy. This is reflected in the widespread use of 30 ng/mL as the boundary of adequacy.
If one is maintaining 25(OH)D this high, one could mimic the maintenance dose of vitamin D used in this study by taking 7,600 IU per day upon the onset of symptoms through recovery.
If one goes into an illness with 25(OH)D below 20 ng/mL, one could quickly bring this level up using a bolus dosing approach like in the RCT. Their approach would equate to using 100,000 IU on the first day, followed by 18,000 IU per day for the next six days, then switching to the maintenance dose; or by using 30,000 IU each day for the first seven days, then switching to the maintenance dose; or by using 100,000 IU on the first day, 50,000 IU on the third day, 50,000 IU on the seventh day, and then switching to the maintenance dose.
Version 6 of the Food and Supplement Guide for the Coronavirus
I have now released Version 6 of The Food and Supplement Guide for the Coronavirus to reflect the new study on vitamin D. Purchases of the guide are greatly appreciated, as they help sustain my work on this newsletter and will help me start finishing my Vitamins and Minerals 101 book.
The Bottom Line
The data are now in. The effect of vitamin D on COVID-19 severity, and likely mortality, is causal. Maintaining 25(OH)D 30-40 ng/mL is likely to be strongly protective against having a severe or fatal case. Use of bolus dosing as described in the “taking action” section upon the first sign of symptoms, if one has levels much lower than this, and otherwise supplementing with a maintenance dose of 7-8,000 IU per day during illness, is likely to be strongly protective against severe and fatal cases.
Stay safe and healthy,
Chris
This is the study in question:
"Effect of Calcifediol Treatment and best Available Therapy versus best Available Therapy on Intensive Care Unit Admission and Mortality Among Patients Hospitalized for COVID-19: A Pilot Randomized Clinical study" - ScienceDirect
Highlights
•
The vitamin D endocrine system may have a variety of actions on cells and tissues involved in COVID-19 progression.
•
Administration of calcifediol or 25-hydroxyvitamin D to hospitalized COVID-19 patients significantly reduced their need for Intensive Care United admission.
•
Calcifediol seems to be able to reduce severity of the disease.
Abstract
Objective
The vitamin D endocrine system may have a variety of actions on cells and tissues involved in COVID-19 progression especially by decreasing the Acute Respiratory Distress Syndrome. Calcifediol can rapidly increase serum 25OHD concentration. We therefore evaluated the effect of calcifediol treatment, on Intensive Care Unit Admission and Mortality rate among Spanish patients hospitalized for COVID-19.
Design
parallel pilot randomized open label, double-masked clinical trial.
Setting
university hospital setting (Reina Sofia University Hospital, Córdoba Spain.)
Participants
76 consecutive patients hospitalized with COVID-19 infection, clinical picture of acute respiratory infection, confirmed by a radiographic pattern of viral pneumonia and by a positive SARS-CoV-2 PCR with CURB65 severity scale (recommending hospital admission in case of total score > 1).
Procedures
All hospitalized patients received as best available therapy the same standard care, (per hospital protocol), of a combination of hydroxychloroquine (400 mg every 12 hours on the first day, and 200 mg every 12 hours for the following 5 days), azithromycin (500 mg orally for 5 days. Eligible patients were allocated at a 2 calcifediol:1 no calcifediol ratio through electronic randomization on the day of admission to take oral calcifediol (0.532 mg), or not. Patients in the calcifediol treatment group continued with oral calcifediol (0.266 mg) on day 3 and 7, and then weekly until discharge or ICU admission. Outcomes of effectiveness included rate of ICU admission and deaths.
Results
Of 50 patients treated with calcifediol, one required admission to the ICU (2%), while of 26 untreated patients, 13 required admission (50%) p value X2 Fischer test p < 0.001. Univariate Risk Estimate Odds Ratio for ICU in patients with Calcifediol treatment versus without Calcifediol treatment: 0.02 (95%CI 0.002-0.17). Multivariate Risk Estimate Odds Ratio for ICU in patients with Calcifediol treatment vs Without Calcifediol treatment ICU (adjusting by Hypertension and T2DM): 0.03 (95%CI: 0.003-0.25). Of the patients treated with calcifediol, none died, and all were discharged, without complications. The 13 patients not treated with calcifediol, who were not admitted to the ICU, were discharged. Of the 13 patients admitted to the ICU, two died and the remaining 11 were discharged.
Conclusion
Our pilot study demonstrated that administration of a high dose of Calcifediol or 25-hydroxyvitamin D, a main metabolite of vitamin D endocrine system, significantly reduced the need for ICU treatment of patients requiring hospitalization due to proven COVID-19. Calcifediol seems to be able to reduce severity of the disease, but larger trials with groups properly matched will be required to show a definitive answer.
Keywords
COVID-19
1. Introduction
A new coronavirus-induced pneumonia was called coronavirus disease 2019 (COVID-19) by the World Health Organization (WHO) on the February 11, 2020, at the same time the international virus classification commission announced that the new coronavirus was named coronavirus 2 severe acute respiratory syndrome (SARS-CoV-2) [1]. Its epidemic spread has increased since it appeared. On the 31 st of January 2020, the WHO announced that COVID-19 was labeled as Public Health Emergency of International Concern (PHEIC).
Patients with COVID-19 show clinical clusters of severe respiratory illness manifestations including fever, nonproductive cough, dyspnea, myalgia, fatigue, abnormal leukocyte counts, and radiographic evidence of pneumonia, which are similar to the symptoms of previous SARS-CoV and MERS-CoV infections [2].
SARS-CoV-2 infection can remain asymptomatic or cause modest symptoms. Severely sick patients require hospital admission and about 20 % of hospitalized patients will developed Acute Respiratory Distress Syndrome (ARDS) and require intensive care unit (ICU) treatment [3]. ARDS, also in patients with Coronavirus Disease 2019 (COVID‐19) is a life‐threatening condition [4,5]. Although frequencies vary according to series, more than 40 % of patients hospitalized because of COVID‐19 pneumonia developed ARDS of which more than 50 % ultimately died [6]. ARDS onset is often rapidly progressive and appears approximately nine days after the onset of severe COVID‐19 [2]. The epidemiologic, morbidity and mortality patterns of ARDS are similar regardless of the trigger [7]. Moreover, ARDS is a pivotal component in the development of multiple organ dysfunction and mortality risk [8]. In the absence well documented effective treatments [4], there is a strong interest in identifying a strategy [9] to taper down the severity of COVID-19, as it would reduce the morbidity and maybe mortality and lower the need for the limited ICU health care resources [10].
It has been proposed that the activation of the vitamin D receptor (VDR) signaling pathway may generate beneficial effects in ARDS [11] by decreasing the cytokine/chemokine storm, regulating the renin‑angiotensin system, modulating neutrophil activity and by maintaining the integrity of the pulmonary epithelial barrier, stimulating epithelial repair and tapering down the increased coagulability [12,13,14,15,16]. Recently, two ecological studies have reported inverse correlations between national estimates of vitamin D status and the incidence and mortality of IDOC-19 in European countries [17,18]; lower concentrations of circulating 25 (OH) D have also been reported to be associated with susceptibility to SARS-CoV-2 infection [19] and the severity of the evolution of COVID-19 [20]. Vitamin D deficiency is frequent in wintertime even in Southern Spain [21] and even more so in patients requiring ICU treatment [22].
Therefore, considering the number of deaths associated to COVID-19, especially the speed with which ARDS is established in a significant number of patients, we performed a pilot study to assess the clinical effectiveness of treatment of patients hospitalized for COVID-19 with calcifediol (25-hydroxyvitamin D3) in early stages to evaluate whether such treatment can reduce the need for admission to ICU and consequently the derived potential risk of death, as a preliminary step to a more extensive randomized clinical trial.
2. Methods
The study protocol was approved by the Pharmacy Committee, and by Ethics committee for the Treatment of COVID-19 of the Reina Sofía University Hospital, Córdoba, Spain EU. (Act-29/2020). The study was conducted in accordance with the principles of the Declaration of Helsinki and the Good Clinical Practice guidelines of the International Conference on Harmonization. All patients and/or legal representatives were verbally informed about the objectives of the trial and their participation, by formally obtaining their consent, and its acceptance recorded in the electronic medical record of the Hospital.
2.1. Study Design Site and participants
Pilot Covidiol was a parallel pilot randomized open label, double-masked clinical study aiming to assess whether calcifediol can reduce the need for admission to ICU, and related death, as a previous part of the clinical trial Covidiol ( Prevention and treatment with Calcifediol of Coronavirus induced acute respiratory syndrome (SARS) COVID-19 (COVIDIOL)” (NCT04366908)) and facilitate the sample calculation. This pilot trial was conducted at Reina Sofia University Hospital, Cordoba Spain.
Were included in the study seventy-sixth consecutive patients hospitalized with COVID-19 infection clinical [23][24] picture of acute respiratory infection, confirmed by a radiographic pattern of viral pneumonia and by a positive SARS-CoV-2 PCR with CURB65 severity scale (recommending hospital admission in case of total score > 1) [25]. Patients younger than 18 years and pregnant women were not included (Fig. 1).
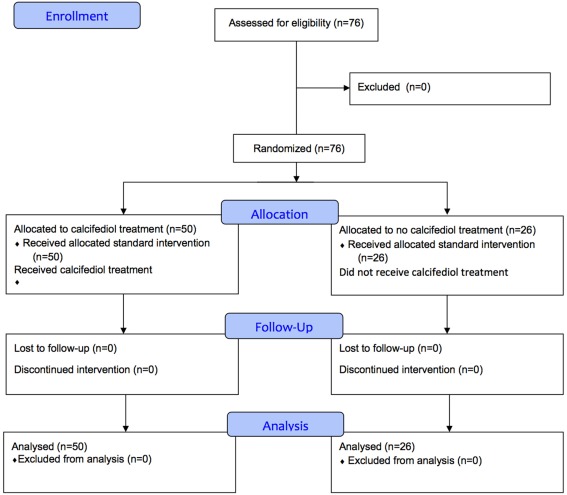
Fig. 1. Patients Flow Diagram.
All hospitalized patients received as best available therapy the same standard care, (per hospital protocol), of a combination of hydroxychloroquine (400 mg every 12 hours on the first day, and 200 mg every 12 hours for the following 5 days), azithromycin (500 mg orally for 5 days) and for patients with pneumonia and NEWS score≥5, a broad spectrum antibiotic (ceftriaxone2 g intravenously every 24 hours for 5 days) was added to hydroxychloroquine and azithromycin.
Hydroxychloroquine (EC50 = 0.72 μM) was chosen because it was in vitro more potent than chloroquine (EC50 = 5.47 μM). Based on physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) models results, a loading dose of 400 mg twice daily of hydroxychloroquine sulfate given orally, followed by a maintenance dose of 200 mg given twice daily for 4 days is recommended for SARS-CoV-2 infection, as it reached 3 times the potency of chloroquine phosphate when given 500 mg twice daily 5 days in advance [26].
The patients were admitted to the ICU by applying the rigorous protocol of the Reina Sofia University Hospital (see supplementary material). Several fundamental aspects were considered when evaluating admission to the ICU: Presence of comorbidities, either individually or quantified in the modified age Charlson Comorbidity Index; Barthel's Index for functional assessment. It establishes the level of dependence of a patient according to his or her needs and clinical criteria: CURB-65 and SOFA scale and ATS/IDSA criteria [27]. A multidisciplinary Selection Committee was created, made up of intensivists, pulmonologists, internists and members of the ethics committee who decided on admission to the ICU.
Sample Size Calculation was carried out for a pilot study with 75 patients randomized in the proportion of 2:1 to carry out the definitive trial (COVIDIOL) (NCT04366908). The sample size calculation is based on the proportion of a participant treated with Calcifediol could meet the criteria for admission to the Intensive Care Unit which is estimated as 5% (with 90% confidence intervals) and the proportion of a participant not treated with Calcifediol which could be 10%. According to these assumptions the estimated final sample size for our pilot clinical study was 50 patients in the arm of patients treated with Calcifediol and 25 patients in the group of patients not treated with Calcifediol [28]. The attrition rate is assumed to be 12%.
2.2. Procedures
Eligible patients were allocated at a 2 calcifediol:1 no calcifediol ratio through electronic randomization performed by hospital statisticians (Fig. 1) on the day of admission to take oral Calcifediol (Faes-Farma, Lejona, Spain), in soft capsules (0.532 mg), or not. Patients in the calcifediol treatment group continued with oral calcifediol (0.266 mg) on day 3 and 7, and then weekly until discharge or ICU admission [22][29]. Patients were followed-up until admission to ICU, hospital discharge or death.
2.2.1. Randomization and Masking
An electronically generated randomization 2:1 list was prepared by independent statisticians. The list was accessible only to nonmasked specialists in the study in an attempt to minimize observation bias. The patients' data were recorded in the hospital's electronic medical record, with blind access by the technical data collectors and the statistician who carried out the study.
2.2.2. Outcomes
Outcomes of effectiveness included rate of ICU admission and deaths. The working hypothesis of this pilot trial was that calcifediol treatment would decrease the need for ICU admissions and the potential risk of death associated with these admissions.
2.3. Laboratory Analysis and respiratory function test
Clinical samples for SARS-CoV-2 diagnostic testing were obtained according to WHO guidelines [30]. For each patient, a sampling strategy was implemented in which samples were obtained on admission. Upper respiratory tract samples were obtained by nasopharyngeal exudate sampling. Procedures for RNA extraction and real-time RT-PCR (rtRT-PCR) were undertaken in the local Central Microbiology Laboratory (Code 202 MagCore® Viral Nucleic Acid Extraction Kit and Allplex™ 2019-nCoV Assay by Seegene or VIASURE SARS-CoV-2 Real Time PCR Detection Kit).
Hematology analyses included blood count (Flow cytometry on ADVIA 2120i, Siemens Healthineers, Erlangen, Germany) and coagulation study including D-Dimer (clotting and immunoturbidimetric assay on ACL TOP 700, Instrumentation Laboratory/Werfen). Biochemical tests including renal function, liver function, lactate dehydrogenase (spectrophotometric assay on Advia chemistry 2400 XPT, Siemens Healthineers, Erlangen, Germany), ferritin and C-reactive protein (immunoturbidimetric assay on Advia chemistry 2400 XPT, Siemens Healthineers, Erlangen, Germany. IL-6 (chemiluminescent immuno assay on Advia Centaur XPT, Siemens Healthineers, Erlangen, Germany)
Respiratory function was assessed by PaO2/FiO2 index [5]. A chest X-ray was taken in all patients on admission All X-ray tests were evaluated by an expert team of chest radiologist.
2.4. Statistical Analysis
Descriptive statistics were used for demographic, laboratory, and clinical prognostic factors related to COVID-19 for each treatment arm.
The comparison between groups of quantitative variables were performed by using t-test for qualitative variables, χ2 tests and Fisher exact tests (with frequencies <5) were used.
Univariate and multivariate logistic regressions were used to estimate Odds ratio and 95% CIs for the probability of admission to ICU. Significant p-value was considered when p < 0.05.
All the analysis has been done using IBM SPSS Statistics software (SPSS).
The pilot trial was reported according to the Consolidated Standards of Reporting Trials (CONSORT) reporting guideline [31].
3. Results
Table 1 shows demographic characteristics of the patients in both groups. Seventy-six patients (45 men (59%) and 31 women) were enrolled in the study and randomized: 26 without calcifediol treatment, 50 with calcifediol treatment (Fig. 1). Mean age was 53 ± 10 (mean ± SD) years, being 54 ± 9 years for men and 51 ± 11 years for women. There was no significant gender difference in age between patients in each group (p = 0.09).
Table 1. Demographic characteristics.
Group receiving Calcifediol (n = 50) Group without Calcifediol (n = 26) IC 95% P
Age (years) 53.14 +/- 10.77 52.77 +/- 9.35 -0.34 – 9.60 0.07
Males [n (%)] 27 (54%) 18 (69%) -0.38 – 0.07 0.20
Females [n (%)] 23 (46%) 8 (31%) -0.07 – 0.38 0.20
Male´s age (years) 56.30 +/ 8.29 52.13 +/- 10.05 -9.67 – 1.41 0.14
Female´s age (years) 49.43 +/- 12.28 54.13+/- 7.99 -4.87 – 14.25 0.32
Results are expressed as mean +/- Standard Deviation.
Baseline factors associated with bad prognosis of COVID-19 are listed in Table 2 as absolute and relative frequencies for categorical variables, and as median plus standard deviation for numerical variables. In addition, both groups were compared for homogeneity at baseline.
Table 2. Prognostic factors for COVID-19 at baseline.
Poor prognosis risk factor Group receiving Calcifediol (n = 50) Group without Calcifediol (n = 26) IC 95% P
≥ 60 years 14 (28%) 5 (19.23%) -0.11 – 0.28 0.40
Previous lung disease 4 (8%) 2 (7.69%) ´-0.12 – 0.13 0.96
Previous Chronic kidney disease 0 0 - -
Previous Diabetes mellitus 3 (6%) 5 (19.23%) -0.30 – 0.03 0.08
Previous High blood pressure 11 (24.19%) 15 (57.69%) -0.58 – -0.13 0.002
Previous Cardiovascular disease 2 (4%) 1 (3.85%) -0.09 – 0.09 0.97
Immunosuppressed & transplanted 6 (12%) 1 (3.85%) -0.03 – 0.20 0.24
At least one prognostic bad risk factora 24 (48%) 16 (61.54%) -0.37 – 0,10
0.26
PaO2/FiO2 (mean +/-SD) 346.57 +/- 73.38 334.62 +/- 66.33 -22.29 – 46.19 0.49
C-reactive protein (mg/L) (mean +/-SD) 82.93 +/- 62.74 94.71 +/- 63.64 -42.15 – 18.59 0.44
LDH (U/L)(mean +/-SD) 308.12 +/- 83.83 345.81 +/- 108.57 -82.46 – 7.08 0.10
D-Dimer (ng/mL) (mean +/-SD) 650.92 +/- 405.61 1333.54 +/- 2570.50 -360.29 – 1725.53 0.19
Lymphocytes < 800/µL 10 (20%) 6 (23.08%) -0.16 – 0.23 0.75
Ferritin (ng/mL) (mean +/-SD) 691.04 +/- 603.54 825.16 +/- 613.95 -166.31 – 434.55 0.36
IL-6 (22/48) (pg/mL) (mean +/-SD)
28.88 +/- 75.05 19.54 +/- 19.45 -41.88 – 23.19 0.41
SD: Standard Deviation.
a
Patients with at least one of the following risk factors (age >60, previous lung disease, chronic kidney disease, diabetes mellitus, hypertension, cardiovascular disease or Immunosuppressed and transplanted patients).
At baseline, there was no significant difference in number of subjects with at least one risk factor. Patients assigned to calcifediol were slightly (not significantly) older, whereas the control group had a higher percentage of hypertension (Table 2).
Among 26 patients not treated with calcifediol, thirteen required ICU admission (50%), while out of fifty patients treated with calcifediol only one required admission to the ICU, whereas the other patients remained in conventional hospitalization
Although at baseline, there was no significant difference in number of subjects with at least one risk factor, the randomization did not achieve a homogeneous distribution of all the variables investigated between the two comparison groups (with and without calcifediol (Table2). A statistically significant difference was identified for the variable hypertension (26 had a history of hypertension of which 11 (42%) received Calcifediol and 15 (58%) not (CI: −0.58 to −0.13; p: 0.002) and close to statistical significance for diabetes 3 (6%) versus 5 (19% ). Therefore, a multivariate logistic regression analysis was performed to adjust the model by possible confounding variables such as hypertension and type 2 diabetes mellitus for the probability of the admission to the Intensive Care Unit in patients with Calcifediol treatment vs Without Calcifediol treatment (odds ratio: 0.03 (95%CI: 0.003-0.25) (Table 3). The dependent variable considered was the need to be treated or not in ICU (dichotomous variable).) CI:-0.30 - 0.03 p:0.08.
Table 3. Requirements for admission to the Intensive Care Unit, in patients hospitalized with COVID-19 (treated or not with calcifediol).
Without Calcifediol Treatment (n = 26) With Calcifediol Treatment (n = 50) p value (1d712;2) Fischer Test
Need for ICU
<0.001
Not requiring ICU, n (%) 13 (50) 49 (98)
Requiring ICU, n (%) 13 (50) 1 (2)
* Univariate Risk Estimate Odds Ratio for ICU in patients with Calcifediol treatment vs Without Calcifediol treatment: 0.02 (95%CI 0.002-0.17).
** Multivariate Risk Estimate Odds Ratio for ICU in patients with Calcifediol treatment vs Without Calcifediol treatment ICU (adjusting by Hypertension and T2DM): 0.03 (95%CI: 0.003-0.25).
Of the patients treated with calcifediol, none died, and all were discharged, without complications. The 13 patients not treated with calcifediol, who were not admitted to the ICU, were discharged. Of the 13 patients admitted to the ICU, two died and the remaining 11 were discharged.
4. Discussion
In line with our hypothesis on a possible link between VDR activation and the severity of ARDS or COVID-19 [11], our pilot study suggests that administration of a high dose of calcifediol or 25-hydroxyvitamin D3, a main metabolite of vitamin D endocrine system, significantly reduced the need for ICU treatment of patients requiring hospitalization due to proven COVID-19.
The best available treatment that at the beginning of the outbreak in our hospital, included the use of hydroxychloroquine/azithromycin therapy [23,24,26]. However, taking into consideration more recent data on the safety and efficacy of chloroquine and hydroxychloroquine in small randomized clinical trials, case series, and observational studies this treatment is no longer considered effective [32] in treating COVID-19.
Randomization generated groups with comparable percentage of unfavorable risk factors as there was no significant difference in subjects with at least one risk factor, except for high blood pressure and diabetes mellitus, known risk factors for unfavorable disease progression [2], which were more frequent in patients not treated with calcifediol.
However, even considering these factors, calcifediol significantly decreased the need for ICU admission in COVID-19 patients in a way not previously reported in this process until now [4]. From a mechanistic perspective there are good reasons to postulate that vitamin D endocrine system favorably modulates host responses to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), both in the later hyperinflammatory and early viraemic phases of COVID-19. as outlined in our previous review [11].
It is important to highlight that the cuboidal alveolar coating cells type II (ACII), like the cells of the immune system express all the enzymatic endowment (see above), to use calcifediol as substrate synthesize 1.25 (OH)2D3 or calcitriol [33]. With high basal expression of 1α-hydroxylase activation and low expression of inactivating enzyme (24-hydroxylase).The result is that ACII constitutively convert calcifediol to 1,25-dihydroxyvitamin D3, the hormonal form of the endocrine system of vitamin D. The calcitriol generated by the ACII acting on themselves and cells of the immune system may then lead to increased expression of genes with important innate immune functions (the antimicrobial cathelicidin peptide, defensins and the TLR co-receptor CD14 etc…). In addition, in a viral infection model, dsRNA leads to increased regulation of 1α-hydroxylase and synergizes with calcifediol and calcitriol sequentially to induce cathelicidin [34].
It should be noted that the role of calcifediol and calcitriol in the animal model and ACII cells [12,34] and the immune system [35,36] were about equipotent suggesting that ACII cells actively converted calcifediol. Interestingly, when ACII cells are treated with a concentration of calcifediol ≥ 10 −7 M (or ≥40 ng / ml) the same effects are achieved as when calcitriol is used, which is a guide to the serum levels of 25OHD3 to be achieved in our trial [12,34].
This pilot study has several limitations as it is not double-blind placebo controlled. On the other hand, in the first studies evaluating risk factors for severe disease and/or death from COVID-19, the possible role of obesity was not considered. Therefore, given the isolation characteristics of the patients, we did not collect the BMI, which would have allowed us to add obesity as a risk factor for severe evolution of COVID-19 [37] It is striking to consider that obesity shares with aging and black or asian ethnicity a surprising overlap as risk factors for severe COVID-19 and vitamin D deficiency [38].
Serum 25OHD concentrations at baseline or during treatment are not available [39,40]. Overall, adults living in the Córdoba area are relatively vitamin D deficient (16 ng/ml on average) in late winter and early spring [17]. Patients with severe ARDS [28,29] or requiring ICU [30] are [17] frequently severely vitamin D deficient. In addition, low serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalized with COVID-19 are associated with greater disease severity [20].
Furthermore, to correct vitamin D deficiency in severely sick patients much higher doses of vitamin D than usual are needed. Our study does not include a comparison with cholecalciferol, the native vitamin D3 form and nutritional substrate for calcifediol, so that we cannot conclude that calcifediol is superior to vitamin D itself. Nevertheless, calcifediol may have some advantages over native vitamin D. It has a more reliable intestinal absorption (close to 100%) and can rapidly restore serum concentrations of 25OHD as it does not require hepatic 25-hydroxylation. This is especially relevant in clinical situations whereby rapid restoration of serum 25OHD is desirable and CYP2R1 expression is compromised. Such impaired CYP2R1 activity has been well demonstrated in several animal models [41] and has also been observed in patients with COPD or asthma [42]. In addition, calcifediol is more potent when compared to oral vitamin D3 [43]. In subjects with a deficient state of vitamin D, and administering physiological doses (up to 25 µg or 1000 IU daily, approximately 1 in 3 molecules of vitamin D appears as 25OHD; the efficacy of conversion is lower (about 1 in 10 molecules) when pharmacological doses of vitamin D/25OHD are used.[42]
The tissue effects of restoring the activation of the vitamin D receptor (VDR) signaling pathway may be due to circulating endocrine 1,25(OH)2D or, more likely, on the local conversion (para/autocrine) of 25OHD into the active hormone in pulmonary alveolar cells, immune cells or other potential target tissues [33].
5. Conclusions
In conclusion, our pilot study demonstrated that administration of calcifediol may improve the clinical outcome of subjects requiring hospitalization for COVID-19. Whether that would also apply to patients with an earlier stage of the disease and whether baseline vitamin D status modifies these results is unknown. Therefore, a multicenter randomized controlled trial using calcifediol, properly matched (Prevention and Treatment With Calcifediol of COVID-19 Induced Acute Respiratory Syndrome (COVIDIOL)), in 15 Spanish hospitals, funded by “Clinical Research Program at COVID-19 “Progreso y Salud” Foundation and Foundation for Biomedical Research of Córdoba (FIBICO), Spain, (registered as NCT04366908 in NIH Trialnet database) will be carried out with the number of patients recalculated from the data provided by this study.
An interesting perspective of the new COVIDIOL trial with the recently available information, could be to evaluate calcifediol associated to dexamethasone or other corticoid vs. dexamethasone or other corticosteroid, since dexamethasone, which has potent anti-inflammatory actions, has recently been shown to reduce mortality in hospitalized patients on Covid-19 who are on respiratory assistance [44]; so that treatment guidelines have been updated to recommend the use of glucocorticoids (including dexametasone) [45], now proposed as the best available treatment in many hospitals around the world
Acknowledgements
Jose Manuel Quesada Gomez y Luis Manuel Entrenas costa had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis". We acknowledge the dedication, commitment, and sacrifice of the staff, and personnel in our institution through the Covid-19 crisis and the suffering and loss of our patients as well as in their families and our Community. We appreciate the statistical work of Ipek Guler, and Carmen Molina. We are grateful to COVID-011-2020 “Programa de Investigación clínica en COVID-19 de Andalucía”. Consejería de Salud y Familia. “Fundación Progreso y Salud” and “Fundación para la Investigación Biomédica de Córdoba” (FIBICO). Andalucía. Spain.
References
[1]
P. Zhou, X. Lou Yang, X.G. Wang, B. Hu, L. Zhang, W. Zhang, H.R. Si, Y. Zhu, B. Li, C.L. Huang, H.D. Chen, J. Chen, Y. Luo, H. Guo, R. Di Jiang, M.Q. Liu, Y. Chen, X.R. Shen, X. Wang, X.S. Zheng, K. Zhao, Q.J. Chen, F. Deng, L.L. Liu, B. Yan, F.X. Zhan, Y.Y. Wang, G.F. Xiao, Z.L. ShiA pneumonia outbreak associated with a new coronavirus of probable bat origin
Nature., 579 (2020), pp. 270-273, 10.1038/s41586-020-2012-7
CrossRefView Record in ScopusGoogle Scholar
[2]
C. Huang, Y. Wang, X. Li, L. Ren, J. Zhao, Y. Hu, L. Zhang, G. Fan, J. Xu, X. Gu, Z. Cheng, T. Yu, J. Xia, Y. Wei, W. Wu, X. Xie, W. Yin, H. Li, M. Liu, Y. Xiao, H. Gao, L. Guo, J. Xie, G. Wang, R. Jiang, Z. Gao, Q. Jin, J. Wang, B. CaoClinical features of patients infected with 2019 novel coronavirus in Wuhan, China
Lancet., 395 (2020), pp. 497-506, 10.1016/S0140-6736(20)30183-5
ArticleDownload PDFView Record in ScopusGoogle Scholar
[3]
N. Chen, M. Zhou, X. Dong, J. Qu, F. Gong, Y. Han, Y. Qiu, J. Wang, Y. Liu, Y. Wei, J. Xia, T. Yu, X. Zhang, L. ZhangEpidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study
Lancet., 395 (2020), pp. 507-513, 10.1016/S0140-6736(20)30211-7
ArticleDownload PDFView Record in ScopusGoogle Scholar
[4]
E. Fan, D. Brodie, A.S. SlutskyAcute Respiratory Distress Syndrome
JAMA., 319 (2018), p. 698, 10.1001/jama.2017.21907
View Record in ScopusGoogle Scholar
[5]
V.M. Ranieri, G.D. Rubenfeld, B.T. Thompson, N.D. Ferguson, E. Caldwell, E. Fan, L. Camporota, A.S. SlutskyAcute respiratory distress syndrome: The Berlin definition
JAMA - J. Am. Med. Assoc., 307 (2012), pp. 2526-2533, 10.1001/jama.2012.5669
View Record in ScopusGoogle Scholar
[6]
C. Wu, X. Chen, Y. Cai, J. Xia, X. Zhou, S. Xu, H. Huang, L. Zhang, X. Zhou, C. Du, Y. Zhang, J. Song, S. Wang, Y. Chao, Z. Yang, J. Xu, X. Zhou, D. Chen, W. Xiong, L. Xu, F. Zhou, J. Jiang, C. Bai, J. Zheng, Y. SongRisk Factors Associated with Acute Respiratory Distress Syndrome and Death in Patients with Coronavirus Disease 2019 Pneumonia in Wuhan, China
JAMA Intern. Med. (2020), 10.1001/jamainternmed.2020.0994
Google Scholar
[7]
G. Bellani, J.G. Laffey, T. Pham, E. Fan, L. Brochard, A. Esteban, L. Gattinoni, F.M.P. Van Haren, A. Larsson, D.F. McAuley, M. Ranieri, G. Rubenfeld, B.T. Thompson, H. Wrigge, A.S. Slutsky, A. PesentiEpidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries
JAMA - J. Am. Med. Assoc., 315 (2016), pp. 788-800, 10.1001/jama.2016.0291
CrossRefView Record in ScopusGoogle Scholar
[8]
Z. Xu, L. Shi, Y. Wang, J. Zhang, L. Huang, C. Zhang, S. Liu, P. Zhao, H. Liu, L. Zhu, Y. Tai, C. Bai, T. Gao, J. Song, P. Xia, J. Dong, J. Zhao, F.S. WangPathological findings of COVID-19 associated with acute respiratory distress syndrome
Lancet Respir. Med., 8 (2020), pp. 420-422, 10.1016/S2213-2600(20)30076-X
ArticleDownload PDFView Record in ScopusGoogle Scholar
[9]
H. Bauchner, P.B. FontanarosaRandomized Clinical Trials and COVID-19: Managing Expectations
JAMA - J. Am. Med. Assoc. (2020), 10.1001/jama.2020.8115
Google Scholar
[10]
R. Li, C. Rivers, Q. Tan, M.B. Murray, E. Toner, M. LipsitchEstimated Demand for US Hospital Inpatient and Intensive Care Unit Beds for Patients With COVID-19 Based on Comparisons With Wuhan and Guangzhou, China
JAMA Netw. Open., 3 (2020), p. e208297, 10.1001/jamanetworkopen.2020.8297
CrossRefView Record in ScopusGoogle Scholar
[11]
J.M. Quesada-Gomez, M. Entrenas-Castillo, R. BouillonVitamin D receptor stimulation to reduce acute respiratory distress syndrome (ARDS) in patients with coronavirus SARS-CoV-2 infections: Revised Ms SBMB 2020_166
J. Steroid Biochem. Mol. Biol., 202 (2020), 10.1016/j.jsbmb.2020.105719
Google Scholar
[12]
Y.Y. Shi, T.J. Liu, J.H. Fu, W. Xu, L.L. Wu, A.N. Hou, X.D. XueVitamin D/VDR signaling attenuates lipopolysaccharide-induced acute lung injury by maintaining the integrity of the pulmonary epithelial barrier
Mol. Med. Rep., 13 (2016), pp. 1186-1194, 10.3892/mmr.2015.4685
CrossRefView Record in ScopusGoogle Scholar
[13]
Y.Y. Shi, T.J. Liu, J.H. Fu, W. Xu, L.L. Wu, A.N. Hou, X.D. XueVitamin D/VDR signaling attenuates lipopolysaccharide-induced acute lung injury by maintaining the integrity of the pulmonary epithelial barrier
Mol. Med. Rep., 13 (2016), pp. 1186-1194, 10.3892/mmr.2015.4685
CrossRefView Record in ScopusGoogle Scholar
[14]
J. Kong, X. Zhu, Y. Shi, T. Liu, Y. Chen, I. Bhan, Q. Zhao, R. Thadhani, Y. Chun LiVDR attenuates acute lung injury by blocking Ang-2-Tie-2 pathway and renin-angiotensin system
Mol. Endocrinol., 27 (2013), pp. 2116-2125, 10.1210/me.2013-1146
CrossRefView Record in ScopusGoogle Scholar
[15]
S.X. Zheng, J.X. Yang, X. Hu, M. Li, Q. Wang, R.C.A. Dancer, D. Parekh, F. Gao-Smith, D.R. Thickett, S.W. JinVitamin D attenuates lung injury via stimulating epithelial repair, reducing epithelial cell apoptosis and inhibits TGF-β induced epithelial to mesenchymal transition
Biochem. Pharmacol., 177 (2020), 10.1016/j.bcp.2020.113955
Google Scholar
[16]
J.M. Martinez-Moreno, C. Herencia, A.M. De Oca, J.R. Muñoz-Castañeda, M.E. Rodríguez-Ortiz, J.M. Diáz-Tocados, E. Peralbo-Santaella, A. Camargo, A. Canalejo, M. Rodriguez, F. Velasco-Gimena, Y. AlmadenVitamin D modulates tissue factor and protease-activated receptor 2 expression in vascular smooth muscle cells
FASEB J., 30 (2016), pp. 1367-1376, 10.1096/fj.15-272872
CrossRefView Record in ScopusGoogle Scholar
[17]
E. Laird, J. Rhodes, R.A. KennyVitamin D. and inflammation: Potential implications for severity of Covid-19
Ir. Med. J., 113 (2020)
Vitamin D and Inflammation: Potential Implications for Severity of Covid-19 - PubMed
(Accessed August 13, 2020)
Google Scholar
[18]
P.C. Ilie, S. Stefanescu, L. SmithThe role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality
Aging Clin. Exp. Res., 32 (2020), pp. 1195-1198, 10.1007/s40520-020-01570-8
CrossRefView Record in ScopusGoogle Scholar
[19]
A. D’avolio, V. Avataneo, A. Manca, J. Cusato, A. De Nicolò, R. Lucchini, F. Keller, M. Cantù25-hydroxyvitamin D concentrations are lower in patients with positive PCR for SARS-CoV-2
Nutrients., 12 (2020), 10.3390/nu12051359
Google Scholar
[20]
G. Panagiotou, S.A. Tee, Y. Ihsan, W. Athar, G. Marchitelli, D. Kelly, C.S. Boot, N. Stock, J. Macfarlane, A.R. Martineau, G. Burns, R. QuintonLow serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalised with COVID-19 are associated with greater disease severity
Clin. Endocrinol. (Oxf). (2020), 10.1111/cen.14276
Google Scholar
[21]
J.M. Mata-Granados, M.D. Luque de Castro, J.M. Quesada GomezInappropriate serum levels of retinol, α-tocopherol, 25 hydroxyvitamin D3 and 24,25 dihydroxyvitamin D3 levels in healthy Spanish adults: Simultaneous assessment by HPLC
Clin. Biochem., 41 (2008), pp. 676-680, 10.1016/j.clinbiochem.2008.02.003
ArticleDownload PDFView Record in ScopusGoogle Scholar
[22]
J.M. Mata-Granados, J. Vargas-Vasserot, C. Ferreiro-Vera, M.D. Luque de Castro, R.G. Pavón, J.M. Quesada GómezEvaluation of vitamin D endocrine system (VDES) status and response to treatment of patients in intensive care units (ICUs) using an on-line SPE-LC-MS/MS method
J. Steroid Biochem. Mol. Biol., 121 (2010), pp. 452-455, 10.1016/j.jsbmb.2010.03.078
ArticleDownload PDFView Record in ScopusGoogle Scholar
[23]
Gobierno de España, Ministerio de Sanidad, Consumo y Bienestar Social - Documentos técnicos para profesionales - Coronavirus (2020)
Ministerio de Sanidad, Consumo y Bienestar Social - Profesionales - Documentos técnicos para profesionales - Coronavirus
(accessed June 22, 2020)
Google Scholar
[24]
Tratamientos disponibles sujetos a condiciones especiales de acceso para el manejo de la infección respiratoria por SARS-CoV-2 - Agencia Española de Medicamentos y Productos Sanitarios, (n.d.). Tratamientos disponibles sujetos a condiciones especiales de acceso para el manejo de la infección respiratoria por SARS-CoV-2 - Agencia Española de Medicamentos y Productos Sanitarios (accessed June 22, 2020).
Google Scholar
[25]
W.S. Lim, M.M. Van Der Eerden, R. Laing, W.G. Boersma, N. Karalus, G.I. Town, S.A. Lewis, J.T. MacfarlaneDefining community acquired pneumonia severity on presentation to hospital: An international derivation and validation study
Thorax., 58 (2003), pp. 377-382, 10.1136/thorax.58.5.377
View Record in ScopusGoogle Scholar
[26]
In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) - PubMed, (n.d.). In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) - PubMed (accessed June 23, 2020).
Google Scholar
[27]
L.A. Mandell, R.G. Wunderink, A. Anzueto, J.G. Bartlett, G.D. Campbell, N.C. Dean, S.F. Dowell, T.M. File, D.M. Musher, M.S. Niederman, A. Torres, C.G. WhitneyInfectious Diseases Society of America/American Thoracic Society Consensus Guidelines on the Management of Community-Acquired Pneumonia in Adults
Clin. Infect. Dis., 44 (2007), pp. S27-S72, 10.1086/511159
CrossRefView Record in ScopusGoogle Scholar
[28]
W. Viechtbauer, L. Smits, D. Kotz, L. Budé, M. Spigt, J. Serroyen, R. CrutzenA simple formula for the calculation of sample size in pilot studies
J. Clin. Epidemiol., 68 (2015), pp. 1375-1379, 10.1016/j.jclinepi.2015.04.014
ArticleDownload PDFView Record in ScopusGoogle Scholar
[29]
S. Russo, L. Carlucci, C. Cipriani, A. Ragno, S. Piemonte, R. Del Fiacco, J. Pepe, V. Fassino, S. Arima, E. Romagnoli, S. MinisolaMetabolic changes following 500 μg monthly administration of calcidiol: A study in normal females
Calcif. Tissue Int., 89 (2011), pp. 252-257, 10.1007/s00223-011-9513-1
CrossRefView Record in ScopusGoogle Scholar
[30]
Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases, (n.d.). Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases (accessed June 23, 2020).
Google Scholar
[31]
K.F. Schulz, D.G. Altman, D. MoherCONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials
BMC Med., 8 (2010), 10.1186/1741-7015-8-18
Google Scholar
[32]
J. Geleris, Y. Sun, J. Platt, J. Zucker, M. Baldwin, G. Hripcsak, A. Labella, D.K. Manson, C. Kubin, R.G. Barr, M.E. Sobieszczyk, N.W. SchlugerObservational study of hydroxychloroquine in hospitalized patients with COVID-19
N. Engl. J. Med., 382 (2020), pp. 2411-2418, 10.1056/NEJMoa2012410
CrossRefView Record in ScopusGoogle Scholar
[33]
R. Bouillon, C. Marcocci, G. Carmeliet, D. Bikle, J.H. White, B. Dawson-Hughes, P. Lips, C.F. Munns, M. Lazaretti-Castro, A. Giustina, J. BilezikianSkeletal and Extraskeletal Actions of Vitamin D: Current Evidence and Outstanding Questions
Endocr. Rev., 40 (2019), pp. 1109-1151, 10.1210/er.2018-00126
CrossRefView Record in ScopusGoogle Scholar
[34]
S. Hansdottir, M.M. Monick, S.L. Hinde, N. Lovan, D.C. Look, G.W. HunninghakeRespiratory Epithelial Cells Convert Inactive Vitamin D to Its Active Form: Potential Effects on Host Defense
J. Immunol., 181 (2008), pp. 7090-7099, 10.4049/jimmunol.181.10.7090
CrossRefView Record in ScopusGoogle Scholar
[35]
A. Rafique, L. Rejnmark, L. Heickendorff, H.J. Møller25(OH)D 3 and 1.25(OH) 2 D 3 inhibits TNF-α expression in human monocyte derived macrophages
PLoS One., 14 (2019), 10.1371/journal.pone.0215383
Google Scholar
[36]
O. Andrukhov, O. Andrukhova, U. Hulan, Y. Tang, H.P. Bantleon, X. Rausch-FanBoth 25-hydroxyvitamin-D3 and 1,25-dihydroxyvitamin- D3 reduces inflammatory response in human periodontal ligament cells
PLoS One, 9 (2014), p. e90301, 10.1371/journal.pone.0090301
CrossRefGoogle Scholar
[37]
A. Simonnet, M. Chetboun, J. Poissy, V. Raverdy, J. Noulette, A. Duhamel, J. Labreuche, D. Mathieu, F. Pattou, M. Jourdain, R. Caizzo, M. Caplan, N. Cousin, T. Duburcq, A. Durand, A. El kalioubie, R. Favory, B. Garcia, P. Girardie, J. Goutay, M. Houard, E. Jaillette, N. Kostuj, G. Ledoux, D. Mathieu, A.S. Moreau, C. Niles, S. Nseir, T. Onimus, E. Parmentier, S. Préau, L. Robriquet, A. Rouze, S. Six, H. VerkindtHigh Prevalence of Obesity in Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) Requiring Invasive Mechanical Ventilation
Obesity., 28 (2020), pp. 1195-1199, 10.1002/oby.22831
CrossRefView Record in ScopusGoogle Scholar
[38]
A.R. Martineau, N.G. ForouhiVitamin D for COVID-19: a case to answer?
Lancet. Diabetes Endocrinol. (2020), 10.1016/S2213-8587(20)30268-0
Google Scholar
[39]
R.P. HeaneyGuidelines for optimizing design and analysis of clinical studies of nutrient effects
Nutr. Rev., 72 (2014), pp. 48-54, 10.1111/nure.12090
CrossRefView Record in ScopusGoogle Scholar
[40]
W.B. Grant, B.J. Boucher, H.P. Bhattoa, H. LahoreWhy vitamin D clinical trials should be based on 25-hydroxyvitamin D concentrations
J. Steroid Biochem. Mol. Biol., 177 (2018), pp. 266-269, 10.1016/j.jsbmb.2017.08.009
ArticleDownload PDFView Record in ScopusGoogle Scholar
[41]
R. Bouillon, D. BikleVitamin D Metabolism Revised: Fall of Dogmas
J. Bone Miner. Res., 34 (2019), pp. 1985-1992, 10.1002/jbmr.3884
CrossRefView Record in ScopusGoogle Scholar
[42]
D.A. Jolliffe, C. Stefanidis, Z. Wang, N.Z. Kermani, V. Dimitrov, J.H. White, J.E. McDonough, W. Janssens, P. Pfeffer, C.J. Griffiths, A. Bush, Y. Guo, S. Christenson, I.M. Adcock, K.F. Chung, K.E. Thummel, A.R. MartineauVitamin D Metabolism is Dysregulated in Asthma and Chronic Obstructive Pulmonary Disease
Am. J. Respir. Crit. Care Med. (2020), 10.1164/rccm.201909-1867oc
Google Scholar
[43]
J.M. Quesada-Gomez, R. BouillonIs calcifediol better than cholecalciferol for vitamin D supplementation?
Osteoporos. Int., 29 (2018), pp. 1697-1711, 10.1007/s00198-018-4520-y
CrossRefView Record in ScopusGoogle Scholar
[44]
RECOVERY Collaborative Group, P. Horby, W.S. Lim, J.R. Emberson, M. Mafham, J.L. Bell, L. Linsell, N. Staplin, C. Brightling, A. Ustianowski, E. Elmahi, B. Prudon, C. Green, T. Felton, D. Chadwick, K. Rege, C. Fegan, L.C. Chappell, S.N. Faust, T. Jaki, K. Jeffery, A. Montgomery, K. Rowan, E. Juszczak, J.K. Baillie, R. Haynes, M.J. LandrayDexamethasone in Hospitalized Patients with Covid-19 - Preliminary Report
N. Engl. J. Med. (2020), 10.1056/NEJMoa2021436
Google Scholar
[45]
Corticosteroids (including dexamethasone). NIH website. Updated July 17, 2020. Accessed August, 2020. Corticosteroids | Coronavirus Disease COVID-19.
Google Scholar
September 3, 2020
The first randomized controlled trial (RCT) of vitamin D in COVID-19 has just been published. The results are astounding: vitamin D nearly abolished the odds of requiring treatment in ICU. Although the number of deaths was too small to say for sure, vitamin D may actually abolish the risk of death from COVID-19.
The First Randomized Controlled Trial on Vitamin D and COVID-19
The trial was conducted at the Reina Sofía University Hospital in Córdoba, Spain. The trial included 76 patients with COVID-19 pneumonia. Although this is no longer the standard of care, all patients were treated with hydroxychloroquine and azithromycin and, when needed, a broad-spectrum antibiotic. Admission to the ICU was determined by a multidisciplinary committee consisting of intensive care specialists, pulmonologists, internal medicine specialists, and members of the ethics committee.
The patients were randomly allocated to receive or not receive vitamin D in a 2:1 ratio. This resulted in 50 patients in the vitamin D group and 26 patients in the control group.
The Vitamin D Treatment Protocol
The vitamin D was provided as oral calcifediol, also known as calcidiol, 25(OH)D, and 25-hydroxyvitamin D. This is a metabolite of vitamin D that our livers make. It is is the principle form of vitamin D that circulates in the blood, and we use it as a measure of vitamin D status.
Traces of 25(OH)D occur in food, and it is five times as potent as vitamin D. As described on page 255 of the 1997 DRI Report, 25(OH)D is given an international unit (IU) value that equates it to vitamin D. Whereas one microgram (mcg) of vitamin D is 40 IU, 1 mcg of 25(OH)D is 200 IU.
The treatment in this RCT was soft capsules of 532 mcg 25(OH)D on day 1 of admission to the hospital, followed by 266 mcg on days 3 and 7, and then 266 mcg once a week until discharge, ICU admission, or death.
This is equivalent to 106,400 IU vitamin D on day 1, 53,200 IU on days 3 and 7, and 53,200 IU weekly thereafter. If this were given as daily doses, it would be the equivalent of 30,400 per day for the first week, followed by a maintenance dose of 7,600 IU per day.
The vitamin D status of the patients was not measured. However, the average vitamin D status in this region of Spain during the time of year in which the study was conducted is 16 ng/mL. A single dose of 100,000 IU vitamin D tends to raise a 25(OH)D of 10 ng/mL into the 20-30 ng/mL range. My suspicion is that the bolus dosing in the first week brought the patients' vitamin D status into the 30-40 ng/mL range by the end of the week, and that most of the healing took place in the circa 40 ng/mL range.
The Results: Near Abolition of ICU Risk
The results are absolutely stunning. 50% of the control group (13 people) required admission to the ICU. Only 2% of those in the vitamin D group (one person) required admission to the ICU.
Expressed as relative risk, vitamin D reduced the risk of ICU admission 25-fold. Put another way, it eliminated 96% of the risk of ICU admission. Expressed as an odds ratio, which is a less intuitive concept but is often used in statistics because it gives an estimate of the effect of the treatment that would be constant across scenarios with different levels of risk, vitamin D reduced the odds of ICU admission by 98%. Either way, vitamin D practically abolished the need for ICU admission.
This was statistically significant at p<0.001, and the 95% confidence interval was 0.002-0.17. This means that the probability of observing differences this large or greater if there is no true effect of vitamin D is less than one in a thousand, and that the probability is 95% that the true effect lies somewhere between an 83% and a 99.8% reduction in the odds of ICU admission.
No matter how you slice it, the effect of vitamin D is extremely compelling.
Because the study is small, the potential confounding variables were not perfectly evenly distributed between the two groups. There was more high blood pressure in the control group, and there was a borderline greater number of patients with diabetes in the control group. Though not statistically significant, the vitamin D group had more people over the age of 60 and five times as many people with organ transplants or who were otherwise on immunosuppressive drugs. To account for all of these differences, they adjusted for them statistically. In the adjusted model, vitamin D still reduced the odds of ICU admission by 97%, with the 95% confidence interval ranging from a 75% to a 99.7% reduction in the odds.
Did Vitamin D Also Abolish the Risk of Death?
All of the vitamin D patients were discharged without complications. Half the control group was discharged without ICU admission. Among the other half, 11 were eventually discharged from ICU, and 2 died.
Although there were, thankfully, too few deaths to run statistics on, since patients generally would be admitted to the ICU before dying, and since vitamin D nearly abolished the risk of ICU admission, we can infer that in a larger study with more deaths, vitamin D would probably abolish or nearly abolish the risk of death.
Comparison With Observational Studies
These results are consistent with the first observational study on vitamin D, which found that 96% of severe and critical cases occurred at 25(OH)D under 30 ng/mL, whereas 97.5% of mild cases had 25(OH)D above 30 ng/mL.
This study couldn't measure the effect on mortality, but all two deaths were in the control group and the near abolition of ICU risk suggests that death would also be nearly abolished. This is consistent with the second observational study finding that only 4% of those with vitamin D status above 30 ng/mL died, while 88% of those with vitamin D status at 20-30 ng/mL died, and 99% of those with vitamin D status lower than that died.
It is less consistent with the weaker observational studies that came out later. For example, the fourth observational study found that prevalence of 25(OH)D below 20 ng/mL differed across three categories of severity judged by CT scan in males but not females. It is also less consistent with more recent studies in Iran and England. In Iran, 25(OH)D below 30 ng/mL only increased risk of severe infection by 21%, although mortality was roughly cut in half at that level. In England, ICU patients were half as likely to have 25(OH)D above 20 ng/mL as non-ICU patients, and mortality wasn't associated with vitamin D status.
In other words, the first RCT shows the effect of vitamin D is closer to the more extreme estimates of the first observational studies than it is to the more moderate estimates of the more recent studies.
The observational studies on infection risk are weaker than those on severity and mortality (see here, here, here, and here), but this RCT didn't look at infection risk.
The RCT contradicts the findings of the two Mendelian randomization studies. These found either no evidence for causality in the vitamin D/COVID-19 connection, or only weak evidence. However, these studies infer causality by looking at the effect of genetics. Genetics only explain 4.2% of the variation in 25(OH)D and many of the genes involved have non-specific, indirect relationships to vitamin D status. This RCT overwhelmingly takes precedence over the findings of the Mendelian randomization studies.
This Study Is the Single Most Important Vitamin D and COVID-19 Study
Since the first vitamin D study was released as a preprint* on April 23, we have been waiting for data that could settle whether the association between vitamin D and COVID-19 incidence, severity, and mortality is a causal one.
This study settles the question: yes, it is causal.
It is important for scientists to replicate each other's findings. Future studies with more diverse populations may reveal differences in the response between different populations. Future larger studies will more precisely refine the exact effect of vitamin D. Future studies with different dosing protocols, in different contexts (for example, without the use of hydroxychloroquine and azithromycin, or with the administration of other drugs or nutrients) will add nuances to our understanding of the effect of vitamin D.
However, given the degree to which it is nearly entirely harmless, and almost without exception beneficial, to maintain 25(OH)D above 30 ng/mL, it would be irresponsible not to interpret this study as definitive evidence of causality. At an absolute minimum, maintaining vitamin D status in this range should be part of the public health message to reduce COVID-19 risk, and 25(OH)D should be universally screened in all hospitals to be treated in anyone with COVID-19, and should be universally screened in all testing centers when antibodies and PCR testing is done, so that everyone knows not only their COVID-19 exposure but also their vitamin D status. If it's low, they should be given advice on how to bring it back up to normal.
How I've Changed My Position
On March 17, when I released Version 1 of The Food and Supplement Guide for the Coronavirus, I expressed concern that vitamin D might increase infection risk or severity by increasing ACE2, the entryway of SARS-CoV-2, the coronavirus that causes COVID-19, into our cells. In response to the first observational study of vitamin D released on April 23, which I covered in this newsletter on April 24, I released version 3 of the guide on April 28, in which I revised this stance and took the position that 25(OH)D should be maintained at 30-34 ng/mL.
As observational studies accumulated, they converged on the common point that 30-40 ng/mL is the sweet spot where infection risk, severity, and mortality are all lowest, without any risk of getting too much vitamin D.
I remained concerned that there might be some risk of a U-shaped curve, where risk increases at vitamin D levels higher than 40 ng/mL. Evidence for or against this was scant and contradictory. Data from Switzerland provided hints of an increased infection risk above 40 in those under 70 years old, but not in those older than 70. Data from Chicago suggested no difference in infection risk above 40 ng/mL, but it didn't separate the people by age. Data from Israel suggested that 25(OH)D above 53 ng/mL completely abolishes infection risk, but everyone with levels that high was under the age of 50. In the Israeli study, the relevant data were only in the young, and risk seemed abolished. In the Swiss study, the relevant data existed for all ages, and it suggested risk increased in the young. All of these studies concern infection risk, not severity or mortality, they all have too little data at high 25(OH)D levels, and they contradict each other.
While I remain agnostic whether there is some level of vitamin D above which infection risk is increased, the new RCT concerns severity and mortality. Although 25(OH)D levels weren't measured, they must have hit at least 40 ng/mL by the time the patients were released, and they possibly exceeded 50 ng/mL. That they gave these patients such massive doses of vitamin D without measuring their vitamin D levels, and that this nearly abolished the need for ICU admission, suggests that there should be no concern about a U-shaped curve with severity or mortality for short-term dosing of up to 8,000 IU per day over several weeks or for temporarily pushing 25(OH)D above 50 ng/mL during the course of treatment.
Given that observational studies around infection risk can consistently show that it is increased at low vitamin D status but cannot show any consistent picture at high vitamin D status, I am, for now, relieving myself of concern about this U-shaped curve. If high vitamin D status can almost eliminate severity and mortality, the off-chance that in some people at some high level it could increase infection risk should be tolerated.
You can see the evolution of my stance on vitamin D through these past posts:
- My Response to Rhonda Patrick on Vitamin D and COVID-19
- Update on Vitamin D and COVID-19 Using the First Observational Study Released
- The Second Study on Vitamin D and COVID-19 Is Now Out
- Two New Vitamin D Studies
- Vitamin D: The First Study on COVID-19 Infection Risk
- The First Vitamin D Study With Pre-Infection Levels Weakens the Association
- Vitamin D Does Not Explain the Race/Ethnicity-COVID-19 Relationship
- Five New Vitamin D and COVID-19 Studies
- The Latest Vitamin D and COVID-19 Study
I think the best way to take action on these findings is to maintain 25(OH)D in 30-40 ng/mL. This is the sweet spot according to the observational literature, and if we assume the patients in the Spanish RCT came in with 16 ng/mL, 30-40 ng/mL probably approximates their average blood level over the course of their recovery.
This is also consistent with other metrics of optimal vitamin D status. All-cause mortality is lowest at about 28 ng/mL according to one meta-analysis of eight European studies, seven conducted in the general population, pooling data from just under 27,000 people. Another widely circulated meta-analysis showing a bottoming-out in the 40-60 ng/mL range derived its data in that range entirely from a conference abstract. If that one outlier is removed, as can clearly be seen in Figure 4 of the paper, the bottoming out of all-cause mortality is below 40 ng/mL and probably close to 30 ng/mL. This is consistent with the amount needed to maximally suppress parathyroid hormone (PTH) in most people, which is the body's own sign of inadequacy in the vitamin D and calcium economy. This is reflected in the widespread use of 30 ng/mL as the boundary of adequacy.
If one is maintaining 25(OH)D this high, one could mimic the maintenance dose of vitamin D used in this study by taking 7,600 IU per day upon the onset of symptoms through recovery.
If one goes into an illness with 25(OH)D below 20 ng/mL, one could quickly bring this level up using a bolus dosing approach like in the RCT. Their approach would equate to using 100,000 IU on the first day, followed by 18,000 IU per day for the next six days, then switching to the maintenance dose; or by using 30,000 IU each day for the first seven days, then switching to the maintenance dose; or by using 100,000 IU on the first day, 50,000 IU on the third day, 50,000 IU on the seventh day, and then switching to the maintenance dose.
Version 6 of the Food and Supplement Guide for the Coronavirus
I have now released Version 6 of The Food and Supplement Guide for the Coronavirus to reflect the new study on vitamin D. Purchases of the guide are greatly appreciated, as they help sustain my work on this newsletter and will help me start finishing my Vitamins and Minerals 101 book.
The Bottom Line
The data are now in. The effect of vitamin D on COVID-19 severity, and likely mortality, is causal. Maintaining 25(OH)D 30-40 ng/mL is likely to be strongly protective against having a severe or fatal case. Use of bolus dosing as described in the “taking action” section upon the first sign of symptoms, if one has levels much lower than this, and otherwise supplementing with a maintenance dose of 7-8,000 IU per day during illness, is likely to be strongly protective against severe and fatal cases.
Stay safe and healthy,
Chris
This is the study in question:
"Effect of Calcifediol Treatment and best Available Therapy versus best Available Therapy on Intensive Care Unit Admission and Mortality Among Patients Hospitalized for COVID-19: A Pilot Randomized Clinical study" - ScienceDirect
Highlights
•
The vitamin D endocrine system may have a variety of actions on cells and tissues involved in COVID-19 progression.
•
Administration of calcifediol or 25-hydroxyvitamin D to hospitalized COVID-19 patients significantly reduced their need for Intensive Care United admission.
•
Calcifediol seems to be able to reduce severity of the disease.
Abstract
Objective
The vitamin D endocrine system may have a variety of actions on cells and tissues involved in COVID-19 progression especially by decreasing the Acute Respiratory Distress Syndrome. Calcifediol can rapidly increase serum 25OHD concentration. We therefore evaluated the effect of calcifediol treatment, on Intensive Care Unit Admission and Mortality rate among Spanish patients hospitalized for COVID-19.
Design
parallel pilot randomized open label, double-masked clinical trial.
Setting
university hospital setting (Reina Sofia University Hospital, Córdoba Spain.)
Participants
76 consecutive patients hospitalized with COVID-19 infection, clinical picture of acute respiratory infection, confirmed by a radiographic pattern of viral pneumonia and by a positive SARS-CoV-2 PCR with CURB65 severity scale (recommending hospital admission in case of total score > 1).
Procedures
All hospitalized patients received as best available therapy the same standard care, (per hospital protocol), of a combination of hydroxychloroquine (400 mg every 12 hours on the first day, and 200 mg every 12 hours for the following 5 days), azithromycin (500 mg orally for 5 days. Eligible patients were allocated at a 2 calcifediol:1 no calcifediol ratio through electronic randomization on the day of admission to take oral calcifediol (0.532 mg), or not. Patients in the calcifediol treatment group continued with oral calcifediol (0.266 mg) on day 3 and 7, and then weekly until discharge or ICU admission. Outcomes of effectiveness included rate of ICU admission and deaths.
Results
Of 50 patients treated with calcifediol, one required admission to the ICU (2%), while of 26 untreated patients, 13 required admission (50%) p value X2 Fischer test p < 0.001. Univariate Risk Estimate Odds Ratio for ICU in patients with Calcifediol treatment versus without Calcifediol treatment: 0.02 (95%CI 0.002-0.17). Multivariate Risk Estimate Odds Ratio for ICU in patients with Calcifediol treatment vs Without Calcifediol treatment ICU (adjusting by Hypertension and T2DM): 0.03 (95%CI: 0.003-0.25). Of the patients treated with calcifediol, none died, and all were discharged, without complications. The 13 patients not treated with calcifediol, who were not admitted to the ICU, were discharged. Of the 13 patients admitted to the ICU, two died and the remaining 11 were discharged.
Conclusion
Our pilot study demonstrated that administration of a high dose of Calcifediol or 25-hydroxyvitamin D, a main metabolite of vitamin D endocrine system, significantly reduced the need for ICU treatment of patients requiring hospitalization due to proven COVID-19. Calcifediol seems to be able to reduce severity of the disease, but larger trials with groups properly matched will be required to show a definitive answer.
Keywords
COVID-19
1. Introduction
A new coronavirus-induced pneumonia was called coronavirus disease 2019 (COVID-19) by the World Health Organization (WHO) on the February 11, 2020, at the same time the international virus classification commission announced that the new coronavirus was named coronavirus 2 severe acute respiratory syndrome (SARS-CoV-2) [1]. Its epidemic spread has increased since it appeared. On the 31 st of January 2020, the WHO announced that COVID-19 was labeled as Public Health Emergency of International Concern (PHEIC).
Patients with COVID-19 show clinical clusters of severe respiratory illness manifestations including fever, nonproductive cough, dyspnea, myalgia, fatigue, abnormal leukocyte counts, and radiographic evidence of pneumonia, which are similar to the symptoms of previous SARS-CoV and MERS-CoV infections [2].
SARS-CoV-2 infection can remain asymptomatic or cause modest symptoms. Severely sick patients require hospital admission and about 20 % of hospitalized patients will developed Acute Respiratory Distress Syndrome (ARDS) and require intensive care unit (ICU) treatment [3]. ARDS, also in patients with Coronavirus Disease 2019 (COVID‐19) is a life‐threatening condition [4,5]. Although frequencies vary according to series, more than 40 % of patients hospitalized because of COVID‐19 pneumonia developed ARDS of which more than 50 % ultimately died [6]. ARDS onset is often rapidly progressive and appears approximately nine days after the onset of severe COVID‐19 [2]. The epidemiologic, morbidity and mortality patterns of ARDS are similar regardless of the trigger [7]. Moreover, ARDS is a pivotal component in the development of multiple organ dysfunction and mortality risk [8]. In the absence well documented effective treatments [4], there is a strong interest in identifying a strategy [9] to taper down the severity of COVID-19, as it would reduce the morbidity and maybe mortality and lower the need for the limited ICU health care resources [10].
It has been proposed that the activation of the vitamin D receptor (VDR) signaling pathway may generate beneficial effects in ARDS [11] by decreasing the cytokine/chemokine storm, regulating the renin‑angiotensin system, modulating neutrophil activity and by maintaining the integrity of the pulmonary epithelial barrier, stimulating epithelial repair and tapering down the increased coagulability [12,13,14,15,16]. Recently, two ecological studies have reported inverse correlations between national estimates of vitamin D status and the incidence and mortality of IDOC-19 in European countries [17,18]; lower concentrations of circulating 25 (OH) D have also been reported to be associated with susceptibility to SARS-CoV-2 infection [19] and the severity of the evolution of COVID-19 [20]. Vitamin D deficiency is frequent in wintertime even in Southern Spain [21] and even more so in patients requiring ICU treatment [22].
Therefore, considering the number of deaths associated to COVID-19, especially the speed with which ARDS is established in a significant number of patients, we performed a pilot study to assess the clinical effectiveness of treatment of patients hospitalized for COVID-19 with calcifediol (25-hydroxyvitamin D3) in early stages to evaluate whether such treatment can reduce the need for admission to ICU and consequently the derived potential risk of death, as a preliminary step to a more extensive randomized clinical trial.
2. Methods
The study protocol was approved by the Pharmacy Committee, and by Ethics committee for the Treatment of COVID-19 of the Reina Sofía University Hospital, Córdoba, Spain EU. (Act-29/2020). The study was conducted in accordance with the principles of the Declaration of Helsinki and the Good Clinical Practice guidelines of the International Conference on Harmonization. All patients and/or legal representatives were verbally informed about the objectives of the trial and their participation, by formally obtaining their consent, and its acceptance recorded in the electronic medical record of the Hospital.
2.1. Study Design Site and participants
Pilot Covidiol was a parallel pilot randomized open label, double-masked clinical study aiming to assess whether calcifediol can reduce the need for admission to ICU, and related death, as a previous part of the clinical trial Covidiol ( Prevention and treatment with Calcifediol of Coronavirus induced acute respiratory syndrome (SARS) COVID-19 (COVIDIOL)” (NCT04366908)) and facilitate the sample calculation. This pilot trial was conducted at Reina Sofia University Hospital, Cordoba Spain.
Were included in the study seventy-sixth consecutive patients hospitalized with COVID-19 infection clinical [23][24] picture of acute respiratory infection, confirmed by a radiographic pattern of viral pneumonia and by a positive SARS-CoV-2 PCR with CURB65 severity scale (recommending hospital admission in case of total score > 1) [25]. Patients younger than 18 years and pregnant women were not included (Fig. 1).

Fig. 1. Patients Flow Diagram.
All hospitalized patients received as best available therapy the same standard care, (per hospital protocol), of a combination of hydroxychloroquine (400 mg every 12 hours on the first day, and 200 mg every 12 hours for the following 5 days), azithromycin (500 mg orally for 5 days) and for patients with pneumonia and NEWS score≥5, a broad spectrum antibiotic (ceftriaxone2 g intravenously every 24 hours for 5 days) was added to hydroxychloroquine and azithromycin.
Hydroxychloroquine (EC50 = 0.72 μM) was chosen because it was in vitro more potent than chloroquine (EC50 = 5.47 μM). Based on physiologically based pharmacokinetic/pharmacodynamic (PBPK/PD) models results, a loading dose of 400 mg twice daily of hydroxychloroquine sulfate given orally, followed by a maintenance dose of 200 mg given twice daily for 4 days is recommended for SARS-CoV-2 infection, as it reached 3 times the potency of chloroquine phosphate when given 500 mg twice daily 5 days in advance [26].
The patients were admitted to the ICU by applying the rigorous protocol of the Reina Sofia University Hospital (see supplementary material). Several fundamental aspects were considered when evaluating admission to the ICU: Presence of comorbidities, either individually or quantified in the modified age Charlson Comorbidity Index; Barthel's Index for functional assessment. It establishes the level of dependence of a patient according to his or her needs and clinical criteria: CURB-65 and SOFA scale and ATS/IDSA criteria [27]. A multidisciplinary Selection Committee was created, made up of intensivists, pulmonologists, internists and members of the ethics committee who decided on admission to the ICU.
Sample Size Calculation was carried out for a pilot study with 75 patients randomized in the proportion of 2:1 to carry out the definitive trial (COVIDIOL) (NCT04366908). The sample size calculation is based on the proportion of a participant treated with Calcifediol could meet the criteria for admission to the Intensive Care Unit which is estimated as 5% (with 90% confidence intervals) and the proportion of a participant not treated with Calcifediol which could be 10%. According to these assumptions the estimated final sample size for our pilot clinical study was 50 patients in the arm of patients treated with Calcifediol and 25 patients in the group of patients not treated with Calcifediol [28]. The attrition rate is assumed to be 12%.
2.2. Procedures
Eligible patients were allocated at a 2 calcifediol:1 no calcifediol ratio through electronic randomization performed by hospital statisticians (Fig. 1) on the day of admission to take oral Calcifediol (Faes-Farma, Lejona, Spain), in soft capsules (0.532 mg), or not. Patients in the calcifediol treatment group continued with oral calcifediol (0.266 mg) on day 3 and 7, and then weekly until discharge or ICU admission [22][29]. Patients were followed-up until admission to ICU, hospital discharge or death.
2.2.1. Randomization and Masking
An electronically generated randomization 2:1 list was prepared by independent statisticians. The list was accessible only to nonmasked specialists in the study in an attempt to minimize observation bias. The patients' data were recorded in the hospital's electronic medical record, with blind access by the technical data collectors and the statistician who carried out the study.
2.2.2. Outcomes
Outcomes of effectiveness included rate of ICU admission and deaths. The working hypothesis of this pilot trial was that calcifediol treatment would decrease the need for ICU admissions and the potential risk of death associated with these admissions.
2.3. Laboratory Analysis and respiratory function test
Clinical samples for SARS-CoV-2 diagnostic testing were obtained according to WHO guidelines [30]. For each patient, a sampling strategy was implemented in which samples were obtained on admission. Upper respiratory tract samples were obtained by nasopharyngeal exudate sampling. Procedures for RNA extraction and real-time RT-PCR (rtRT-PCR) were undertaken in the local Central Microbiology Laboratory (Code 202 MagCore® Viral Nucleic Acid Extraction Kit and Allplex™ 2019-nCoV Assay by Seegene or VIASURE SARS-CoV-2 Real Time PCR Detection Kit).
Hematology analyses included blood count (Flow cytometry on ADVIA 2120i, Siemens Healthineers, Erlangen, Germany) and coagulation study including D-Dimer (clotting and immunoturbidimetric assay on ACL TOP 700, Instrumentation Laboratory/Werfen). Biochemical tests including renal function, liver function, lactate dehydrogenase (spectrophotometric assay on Advia chemistry 2400 XPT, Siemens Healthineers, Erlangen, Germany), ferritin and C-reactive protein (immunoturbidimetric assay on Advia chemistry 2400 XPT, Siemens Healthineers, Erlangen, Germany. IL-6 (chemiluminescent immuno assay on Advia Centaur XPT, Siemens Healthineers, Erlangen, Germany)
Respiratory function was assessed by PaO2/FiO2 index [5]. A chest X-ray was taken in all patients on admission All X-ray tests were evaluated by an expert team of chest radiologist.
2.4. Statistical Analysis
Descriptive statistics were used for demographic, laboratory, and clinical prognostic factors related to COVID-19 for each treatment arm.
The comparison between groups of quantitative variables were performed by using t-test for qualitative variables, χ2 tests and Fisher exact tests (with frequencies <5) were used.
Univariate and multivariate logistic regressions were used to estimate Odds ratio and 95% CIs for the probability of admission to ICU. Significant p-value was considered when p < 0.05.
All the analysis has been done using IBM SPSS Statistics software (SPSS).
The pilot trial was reported according to the Consolidated Standards of Reporting Trials (CONSORT) reporting guideline [31].
3. Results
Table 1 shows demographic characteristics of the patients in both groups. Seventy-six patients (45 men (59%) and 31 women) were enrolled in the study and randomized: 26 without calcifediol treatment, 50 with calcifediol treatment (Fig. 1). Mean age was 53 ± 10 (mean ± SD) years, being 54 ± 9 years for men and 51 ± 11 years for women. There was no significant gender difference in age between patients in each group (p = 0.09).
Table 1. Demographic characteristics.
Group receiving Calcifediol (n = 50) Group without Calcifediol (n = 26) IC 95% P
Age (years) 53.14 +/- 10.77 52.77 +/- 9.35 -0.34 – 9.60 0.07
Males [n (%)] 27 (54%) 18 (69%) -0.38 – 0.07 0.20
Females [n (%)] 23 (46%) 8 (31%) -0.07 – 0.38 0.20
Male´s age (years) 56.30 +/ 8.29 52.13 +/- 10.05 -9.67 – 1.41 0.14
Female´s age (years) 49.43 +/- 12.28 54.13+/- 7.99 -4.87 – 14.25 0.32
Results are expressed as mean +/- Standard Deviation.
Baseline factors associated with bad prognosis of COVID-19 are listed in Table 2 as absolute and relative frequencies for categorical variables, and as median plus standard deviation for numerical variables. In addition, both groups were compared for homogeneity at baseline.
Table 2. Prognostic factors for COVID-19 at baseline.
Poor prognosis risk factor Group receiving Calcifediol (n = 50) Group without Calcifediol (n = 26) IC 95% P
≥ 60 years 14 (28%) 5 (19.23%) -0.11 – 0.28 0.40
Previous lung disease 4 (8%) 2 (7.69%) ´-0.12 – 0.13 0.96
Previous Chronic kidney disease 0 0 - -
Previous Diabetes mellitus 3 (6%) 5 (19.23%) -0.30 – 0.03 0.08
Previous High blood pressure 11 (24.19%) 15 (57.69%) -0.58 – -0.13 0.002
Previous Cardiovascular disease 2 (4%) 1 (3.85%) -0.09 – 0.09 0.97
Immunosuppressed & transplanted 6 (12%) 1 (3.85%) -0.03 – 0.20 0.24
At least one prognostic bad risk factora 24 (48%) 16 (61.54%) -0.37 – 0,10
0.26
PaO2/FiO2 (mean +/-SD) 346.57 +/- 73.38 334.62 +/- 66.33 -22.29 – 46.19 0.49
C-reactive protein (mg/L) (mean +/-SD) 82.93 +/- 62.74 94.71 +/- 63.64 -42.15 – 18.59 0.44
LDH (U/L)(mean +/-SD) 308.12 +/- 83.83 345.81 +/- 108.57 -82.46 – 7.08 0.10
D-Dimer (ng/mL) (mean +/-SD) 650.92 +/- 405.61 1333.54 +/- 2570.50 -360.29 – 1725.53 0.19
Lymphocytes < 800/µL 10 (20%) 6 (23.08%) -0.16 – 0.23 0.75
Ferritin (ng/mL) (mean +/-SD) 691.04 +/- 603.54 825.16 +/- 613.95 -166.31 – 434.55 0.36
IL-6 (22/48) (pg/mL) (mean +/-SD)
28.88 +/- 75.05 19.54 +/- 19.45 -41.88 – 23.19 0.41
SD: Standard Deviation.
a
Patients with at least one of the following risk factors (age >60, previous lung disease, chronic kidney disease, diabetes mellitus, hypertension, cardiovascular disease or Immunosuppressed and transplanted patients).
At baseline, there was no significant difference in number of subjects with at least one risk factor. Patients assigned to calcifediol were slightly (not significantly) older, whereas the control group had a higher percentage of hypertension (Table 2).
Among 26 patients not treated with calcifediol, thirteen required ICU admission (50%), while out of fifty patients treated with calcifediol only one required admission to the ICU, whereas the other patients remained in conventional hospitalization
Although at baseline, there was no significant difference in number of subjects with at least one risk factor, the randomization did not achieve a homogeneous distribution of all the variables investigated between the two comparison groups (with and without calcifediol (Table2). A statistically significant difference was identified for the variable hypertension (26 had a history of hypertension of which 11 (42%) received Calcifediol and 15 (58%) not (CI: −0.58 to −0.13; p: 0.002) and close to statistical significance for diabetes 3 (6%) versus 5 (19% ). Therefore, a multivariate logistic regression analysis was performed to adjust the model by possible confounding variables such as hypertension and type 2 diabetes mellitus for the probability of the admission to the Intensive Care Unit in patients with Calcifediol treatment vs Without Calcifediol treatment (odds ratio: 0.03 (95%CI: 0.003-0.25) (Table 3). The dependent variable considered was the need to be treated or not in ICU (dichotomous variable).) CI:-0.30 - 0.03 p:0.08.
Table 3. Requirements for admission to the Intensive Care Unit, in patients hospitalized with COVID-19 (treated or not with calcifediol).
Without Calcifediol Treatment (n = 26) With Calcifediol Treatment (n = 50) p value (1d712;2) Fischer Test
Need for ICU
<0.001
Not requiring ICU, n (%) 13 (50) 49 (98)
Requiring ICU, n (%) 13 (50) 1 (2)
* Univariate Risk Estimate Odds Ratio for ICU in patients with Calcifediol treatment vs Without Calcifediol treatment: 0.02 (95%CI 0.002-0.17).
** Multivariate Risk Estimate Odds Ratio for ICU in patients with Calcifediol treatment vs Without Calcifediol treatment ICU (adjusting by Hypertension and T2DM): 0.03 (95%CI: 0.003-0.25).
Of the patients treated with calcifediol, none died, and all were discharged, without complications. The 13 patients not treated with calcifediol, who were not admitted to the ICU, were discharged. Of the 13 patients admitted to the ICU, two died and the remaining 11 were discharged.
4. Discussion
In line with our hypothesis on a possible link between VDR activation and the severity of ARDS or COVID-19 [11], our pilot study suggests that administration of a high dose of calcifediol or 25-hydroxyvitamin D3, a main metabolite of vitamin D endocrine system, significantly reduced the need for ICU treatment of patients requiring hospitalization due to proven COVID-19.
The best available treatment that at the beginning of the outbreak in our hospital, included the use of hydroxychloroquine/azithromycin therapy [23,24,26]. However, taking into consideration more recent data on the safety and efficacy of chloroquine and hydroxychloroquine in small randomized clinical trials, case series, and observational studies this treatment is no longer considered effective [32] in treating COVID-19.
Randomization generated groups with comparable percentage of unfavorable risk factors as there was no significant difference in subjects with at least one risk factor, except for high blood pressure and diabetes mellitus, known risk factors for unfavorable disease progression [2], which were more frequent in patients not treated with calcifediol.
However, even considering these factors, calcifediol significantly decreased the need for ICU admission in COVID-19 patients in a way not previously reported in this process until now [4]. From a mechanistic perspective there are good reasons to postulate that vitamin D endocrine system favorably modulates host responses to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), both in the later hyperinflammatory and early viraemic phases of COVID-19. as outlined in our previous review [11].
It is important to highlight that the cuboidal alveolar coating cells type II (ACII), like the cells of the immune system express all the enzymatic endowment (see above), to use calcifediol as substrate synthesize 1.25 (OH)2D3 or calcitriol [33]. With high basal expression of 1α-hydroxylase activation and low expression of inactivating enzyme (24-hydroxylase).The result is that ACII constitutively convert calcifediol to 1,25-dihydroxyvitamin D3, the hormonal form of the endocrine system of vitamin D. The calcitriol generated by the ACII acting on themselves and cells of the immune system may then lead to increased expression of genes with important innate immune functions (the antimicrobial cathelicidin peptide, defensins and the TLR co-receptor CD14 etc…). In addition, in a viral infection model, dsRNA leads to increased regulation of 1α-hydroxylase and synergizes with calcifediol and calcitriol sequentially to induce cathelicidin [34].
It should be noted that the role of calcifediol and calcitriol in the animal model and ACII cells [12,34] and the immune system [35,36] were about equipotent suggesting that ACII cells actively converted calcifediol. Interestingly, when ACII cells are treated with a concentration of calcifediol ≥ 10 −7 M (or ≥40 ng / ml) the same effects are achieved as when calcitriol is used, which is a guide to the serum levels of 25OHD3 to be achieved in our trial [12,34].
This pilot study has several limitations as it is not double-blind placebo controlled. On the other hand, in the first studies evaluating risk factors for severe disease and/or death from COVID-19, the possible role of obesity was not considered. Therefore, given the isolation characteristics of the patients, we did not collect the BMI, which would have allowed us to add obesity as a risk factor for severe evolution of COVID-19 [37] It is striking to consider that obesity shares with aging and black or asian ethnicity a surprising overlap as risk factors for severe COVID-19 and vitamin D deficiency [38].
Serum 25OHD concentrations at baseline or during treatment are not available [39,40]. Overall, adults living in the Córdoba area are relatively vitamin D deficient (16 ng/ml on average) in late winter and early spring [17]. Patients with severe ARDS [28,29] or requiring ICU [30] are [17] frequently severely vitamin D deficient. In addition, low serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalized with COVID-19 are associated with greater disease severity [20].
Furthermore, to correct vitamin D deficiency in severely sick patients much higher doses of vitamin D than usual are needed. Our study does not include a comparison with cholecalciferol, the native vitamin D3 form and nutritional substrate for calcifediol, so that we cannot conclude that calcifediol is superior to vitamin D itself. Nevertheless, calcifediol may have some advantages over native vitamin D. It has a more reliable intestinal absorption (close to 100%) and can rapidly restore serum concentrations of 25OHD as it does not require hepatic 25-hydroxylation. This is especially relevant in clinical situations whereby rapid restoration of serum 25OHD is desirable and CYP2R1 expression is compromised. Such impaired CYP2R1 activity has been well demonstrated in several animal models [41] and has also been observed in patients with COPD or asthma [42]. In addition, calcifediol is more potent when compared to oral vitamin D3 [43]. In subjects with a deficient state of vitamin D, and administering physiological doses (up to 25 µg or 1000 IU daily, approximately 1 in 3 molecules of vitamin D appears as 25OHD; the efficacy of conversion is lower (about 1 in 10 molecules) when pharmacological doses of vitamin D/25OHD are used.[42]
The tissue effects of restoring the activation of the vitamin D receptor (VDR) signaling pathway may be due to circulating endocrine 1,25(OH)2D or, more likely, on the local conversion (para/autocrine) of 25OHD into the active hormone in pulmonary alveolar cells, immune cells or other potential target tissues [33].
5. Conclusions
In conclusion, our pilot study demonstrated that administration of calcifediol may improve the clinical outcome of subjects requiring hospitalization for COVID-19. Whether that would also apply to patients with an earlier stage of the disease and whether baseline vitamin D status modifies these results is unknown. Therefore, a multicenter randomized controlled trial using calcifediol, properly matched (Prevention and Treatment With Calcifediol of COVID-19 Induced Acute Respiratory Syndrome (COVIDIOL)), in 15 Spanish hospitals, funded by “Clinical Research Program at COVID-19 “Progreso y Salud” Foundation and Foundation for Biomedical Research of Córdoba (FIBICO), Spain, (registered as NCT04366908 in NIH Trialnet database) will be carried out with the number of patients recalculated from the data provided by this study.
An interesting perspective of the new COVIDIOL trial with the recently available information, could be to evaluate calcifediol associated to dexamethasone or other corticoid vs. dexamethasone or other corticosteroid, since dexamethasone, which has potent anti-inflammatory actions, has recently been shown to reduce mortality in hospitalized patients on Covid-19 who are on respiratory assistance [44]; so that treatment guidelines have been updated to recommend the use of glucocorticoids (including dexametasone) [45], now proposed as the best available treatment in many hospitals around the world
Acknowledgements
Jose Manuel Quesada Gomez y Luis Manuel Entrenas costa had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis". We acknowledge the dedication, commitment, and sacrifice of the staff, and personnel in our institution through the Covid-19 crisis and the suffering and loss of our patients as well as in their families and our Community. We appreciate the statistical work of Ipek Guler, and Carmen Molina. We are grateful to COVID-011-2020 “Programa de Investigación clínica en COVID-19 de Andalucía”. Consejería de Salud y Familia. “Fundación Progreso y Salud” and “Fundación para la Investigación Biomédica de Córdoba” (FIBICO). Andalucía. Spain.
References
[1]
P. Zhou, X. Lou Yang, X.G. Wang, B. Hu, L. Zhang, W. Zhang, H.R. Si, Y. Zhu, B. Li, C.L. Huang, H.D. Chen, J. Chen, Y. Luo, H. Guo, R. Di Jiang, M.Q. Liu, Y. Chen, X.R. Shen, X. Wang, X.S. Zheng, K. Zhao, Q.J. Chen, F. Deng, L.L. Liu, B. Yan, F.X. Zhan, Y.Y. Wang, G.F. Xiao, Z.L. ShiA pneumonia outbreak associated with a new coronavirus of probable bat origin
Nature., 579 (2020), pp. 270-273, 10.1038/s41586-020-2012-7
CrossRefView Record in ScopusGoogle Scholar
[2]
C. Huang, Y. Wang, X. Li, L. Ren, J. Zhao, Y. Hu, L. Zhang, G. Fan, J. Xu, X. Gu, Z. Cheng, T. Yu, J. Xia, Y. Wei, W. Wu, X. Xie, W. Yin, H. Li, M. Liu, Y. Xiao, H. Gao, L. Guo, J. Xie, G. Wang, R. Jiang, Z. Gao, Q. Jin, J. Wang, B. CaoClinical features of patients infected with 2019 novel coronavirus in Wuhan, China
Lancet., 395 (2020), pp. 497-506, 10.1016/S0140-6736(20)30183-5
ArticleDownload PDFView Record in ScopusGoogle Scholar
[3]
N. Chen, M. Zhou, X. Dong, J. Qu, F. Gong, Y. Han, Y. Qiu, J. Wang, Y. Liu, Y. Wei, J. Xia, T. Yu, X. Zhang, L. ZhangEpidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study
Lancet., 395 (2020), pp. 507-513, 10.1016/S0140-6736(20)30211-7
ArticleDownload PDFView Record in ScopusGoogle Scholar
[4]
E. Fan, D. Brodie, A.S. SlutskyAcute Respiratory Distress Syndrome
JAMA., 319 (2018), p. 698, 10.1001/jama.2017.21907
View Record in ScopusGoogle Scholar
[5]
V.M. Ranieri, G.D. Rubenfeld, B.T. Thompson, N.D. Ferguson, E. Caldwell, E. Fan, L. Camporota, A.S. SlutskyAcute respiratory distress syndrome: The Berlin definition
JAMA - J. Am. Med. Assoc., 307 (2012), pp. 2526-2533, 10.1001/jama.2012.5669
View Record in ScopusGoogle Scholar
[6]
C. Wu, X. Chen, Y. Cai, J. Xia, X. Zhou, S. Xu, H. Huang, L. Zhang, X. Zhou, C. Du, Y. Zhang, J. Song, S. Wang, Y. Chao, Z. Yang, J. Xu, X. Zhou, D. Chen, W. Xiong, L. Xu, F. Zhou, J. Jiang, C. Bai, J. Zheng, Y. SongRisk Factors Associated with Acute Respiratory Distress Syndrome and Death in Patients with Coronavirus Disease 2019 Pneumonia in Wuhan, China
JAMA Intern. Med. (2020), 10.1001/jamainternmed.2020.0994
Google Scholar
[7]
G. Bellani, J.G. Laffey, T. Pham, E. Fan, L. Brochard, A. Esteban, L. Gattinoni, F.M.P. Van Haren, A. Larsson, D.F. McAuley, M. Ranieri, G. Rubenfeld, B.T. Thompson, H. Wrigge, A.S. Slutsky, A. PesentiEpidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries
JAMA - J. Am. Med. Assoc., 315 (2016), pp. 788-800, 10.1001/jama.2016.0291
CrossRefView Record in ScopusGoogle Scholar
[8]
Z. Xu, L. Shi, Y. Wang, J. Zhang, L. Huang, C. Zhang, S. Liu, P. Zhao, H. Liu, L. Zhu, Y. Tai, C. Bai, T. Gao, J. Song, P. Xia, J. Dong, J. Zhao, F.S. WangPathological findings of COVID-19 associated with acute respiratory distress syndrome
Lancet Respir. Med., 8 (2020), pp. 420-422, 10.1016/S2213-2600(20)30076-X
ArticleDownload PDFView Record in ScopusGoogle Scholar
[9]
H. Bauchner, P.B. FontanarosaRandomized Clinical Trials and COVID-19: Managing Expectations
JAMA - J. Am. Med. Assoc. (2020), 10.1001/jama.2020.8115
Google Scholar
[10]
R. Li, C. Rivers, Q. Tan, M.B. Murray, E. Toner, M. LipsitchEstimated Demand for US Hospital Inpatient and Intensive Care Unit Beds for Patients With COVID-19 Based on Comparisons With Wuhan and Guangzhou, China
JAMA Netw. Open., 3 (2020), p. e208297, 10.1001/jamanetworkopen.2020.8297
CrossRefView Record in ScopusGoogle Scholar
[11]
J.M. Quesada-Gomez, M. Entrenas-Castillo, R. BouillonVitamin D receptor stimulation to reduce acute respiratory distress syndrome (ARDS) in patients with coronavirus SARS-CoV-2 infections: Revised Ms SBMB 2020_166
J. Steroid Biochem. Mol. Biol., 202 (2020), 10.1016/j.jsbmb.2020.105719
Google Scholar
[12]
Y.Y. Shi, T.J. Liu, J.H. Fu, W. Xu, L.L. Wu, A.N. Hou, X.D. XueVitamin D/VDR signaling attenuates lipopolysaccharide-induced acute lung injury by maintaining the integrity of the pulmonary epithelial barrier
Mol. Med. Rep., 13 (2016), pp. 1186-1194, 10.3892/mmr.2015.4685
CrossRefView Record in ScopusGoogle Scholar
[13]
Y.Y. Shi, T.J. Liu, J.H. Fu, W. Xu, L.L. Wu, A.N. Hou, X.D. XueVitamin D/VDR signaling attenuates lipopolysaccharide-induced acute lung injury by maintaining the integrity of the pulmonary epithelial barrier
Mol. Med. Rep., 13 (2016), pp. 1186-1194, 10.3892/mmr.2015.4685
CrossRefView Record in ScopusGoogle Scholar
[14]
J. Kong, X. Zhu, Y. Shi, T. Liu, Y. Chen, I. Bhan, Q. Zhao, R. Thadhani, Y. Chun LiVDR attenuates acute lung injury by blocking Ang-2-Tie-2 pathway and renin-angiotensin system
Mol. Endocrinol., 27 (2013), pp. 2116-2125, 10.1210/me.2013-1146
CrossRefView Record in ScopusGoogle Scholar
[15]
S.X. Zheng, J.X. Yang, X. Hu, M. Li, Q. Wang, R.C.A. Dancer, D. Parekh, F. Gao-Smith, D.R. Thickett, S.W. JinVitamin D attenuates lung injury via stimulating epithelial repair, reducing epithelial cell apoptosis and inhibits TGF-β induced epithelial to mesenchymal transition
Biochem. Pharmacol., 177 (2020), 10.1016/j.bcp.2020.113955
Google Scholar
[16]
J.M. Martinez-Moreno, C. Herencia, A.M. De Oca, J.R. Muñoz-Castañeda, M.E. Rodríguez-Ortiz, J.M. Diáz-Tocados, E. Peralbo-Santaella, A. Camargo, A. Canalejo, M. Rodriguez, F. Velasco-Gimena, Y. AlmadenVitamin D modulates tissue factor and protease-activated receptor 2 expression in vascular smooth muscle cells
FASEB J., 30 (2016), pp. 1367-1376, 10.1096/fj.15-272872
CrossRefView Record in ScopusGoogle Scholar
[17]
E. Laird, J. Rhodes, R.A. KennyVitamin D. and inflammation: Potential implications for severity of Covid-19
Ir. Med. J., 113 (2020)
Vitamin D and Inflammation: Potential Implications for Severity of Covid-19 - PubMed
(Accessed August 13, 2020)
Google Scholar
[18]
P.C. Ilie, S. Stefanescu, L. SmithThe role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality
Aging Clin. Exp. Res., 32 (2020), pp. 1195-1198, 10.1007/s40520-020-01570-8
CrossRefView Record in ScopusGoogle Scholar
[19]
A. D’avolio, V. Avataneo, A. Manca, J. Cusato, A. De Nicolò, R. Lucchini, F. Keller, M. Cantù25-hydroxyvitamin D concentrations are lower in patients with positive PCR for SARS-CoV-2
Nutrients., 12 (2020), 10.3390/nu12051359
Google Scholar
[20]
G. Panagiotou, S.A. Tee, Y. Ihsan, W. Athar, G. Marchitelli, D. Kelly, C.S. Boot, N. Stock, J. Macfarlane, A.R. Martineau, G. Burns, R. QuintonLow serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalised with COVID-19 are associated with greater disease severity
Clin. Endocrinol. (Oxf). (2020), 10.1111/cen.14276
Google Scholar
[21]
J.M. Mata-Granados, M.D. Luque de Castro, J.M. Quesada GomezInappropriate serum levels of retinol, α-tocopherol, 25 hydroxyvitamin D3 and 24,25 dihydroxyvitamin D3 levels in healthy Spanish adults: Simultaneous assessment by HPLC
Clin. Biochem., 41 (2008), pp. 676-680, 10.1016/j.clinbiochem.2008.02.003
ArticleDownload PDFView Record in ScopusGoogle Scholar
[22]
J.M. Mata-Granados, J. Vargas-Vasserot, C. Ferreiro-Vera, M.D. Luque de Castro, R.G. Pavón, J.M. Quesada GómezEvaluation of vitamin D endocrine system (VDES) status and response to treatment of patients in intensive care units (ICUs) using an on-line SPE-LC-MS/MS method
J. Steroid Biochem. Mol. Biol., 121 (2010), pp. 452-455, 10.1016/j.jsbmb.2010.03.078
ArticleDownload PDFView Record in ScopusGoogle Scholar
[23]
Gobierno de España, Ministerio de Sanidad, Consumo y Bienestar Social - Documentos técnicos para profesionales - Coronavirus (2020)
Ministerio de Sanidad, Consumo y Bienestar Social - Profesionales - Documentos técnicos para profesionales - Coronavirus
(accessed June 22, 2020)
Google Scholar
[24]
Tratamientos disponibles sujetos a condiciones especiales de acceso para el manejo de la infección respiratoria por SARS-CoV-2 - Agencia Española de Medicamentos y Productos Sanitarios, (n.d.). Tratamientos disponibles sujetos a condiciones especiales de acceso para el manejo de la infección respiratoria por SARS-CoV-2 - Agencia Española de Medicamentos y Productos Sanitarios (accessed June 22, 2020).
Google Scholar
[25]
W.S. Lim, M.M. Van Der Eerden, R. Laing, W.G. Boersma, N. Karalus, G.I. Town, S.A. Lewis, J.T. MacfarlaneDefining community acquired pneumonia severity on presentation to hospital: An international derivation and validation study
Thorax., 58 (2003), pp. 377-382, 10.1136/thorax.58.5.377
View Record in ScopusGoogle Scholar
[26]
In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) - PubMed, (n.d.). In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) - PubMed (accessed June 23, 2020).
Google Scholar
[27]
L.A. Mandell, R.G. Wunderink, A. Anzueto, J.G. Bartlett, G.D. Campbell, N.C. Dean, S.F. Dowell, T.M. File, D.M. Musher, M.S. Niederman, A. Torres, C.G. WhitneyInfectious Diseases Society of America/American Thoracic Society Consensus Guidelines on the Management of Community-Acquired Pneumonia in Adults
Clin. Infect. Dis., 44 (2007), pp. S27-S72, 10.1086/511159
CrossRefView Record in ScopusGoogle Scholar
[28]
W. Viechtbauer, L. Smits, D. Kotz, L. Budé, M. Spigt, J. Serroyen, R. CrutzenA simple formula for the calculation of sample size in pilot studies
J. Clin. Epidemiol., 68 (2015), pp. 1375-1379, 10.1016/j.jclinepi.2015.04.014
ArticleDownload PDFView Record in ScopusGoogle Scholar
[29]
S. Russo, L. Carlucci, C. Cipriani, A. Ragno, S. Piemonte, R. Del Fiacco, J. Pepe, V. Fassino, S. Arima, E. Romagnoli, S. MinisolaMetabolic changes following 500 μg monthly administration of calcidiol: A study in normal females
Calcif. Tissue Int., 89 (2011), pp. 252-257, 10.1007/s00223-011-9513-1
CrossRefView Record in ScopusGoogle Scholar
[30]
Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases, (n.d.). Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases (accessed June 23, 2020).
Google Scholar
[31]
K.F. Schulz, D.G. Altman, D. MoherCONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials
BMC Med., 8 (2010), 10.1186/1741-7015-8-18
Google Scholar
[32]
J. Geleris, Y. Sun, J. Platt, J. Zucker, M. Baldwin, G. Hripcsak, A. Labella, D.K. Manson, C. Kubin, R.G. Barr, M.E. Sobieszczyk, N.W. SchlugerObservational study of hydroxychloroquine in hospitalized patients with COVID-19
N. Engl. J. Med., 382 (2020), pp. 2411-2418, 10.1056/NEJMoa2012410
CrossRefView Record in ScopusGoogle Scholar
[33]
R. Bouillon, C. Marcocci, G. Carmeliet, D. Bikle, J.H. White, B. Dawson-Hughes, P. Lips, C.F. Munns, M. Lazaretti-Castro, A. Giustina, J. BilezikianSkeletal and Extraskeletal Actions of Vitamin D: Current Evidence and Outstanding Questions
Endocr. Rev., 40 (2019), pp. 1109-1151, 10.1210/er.2018-00126
CrossRefView Record in ScopusGoogle Scholar
[34]
S. Hansdottir, M.M. Monick, S.L. Hinde, N. Lovan, D.C. Look, G.W. HunninghakeRespiratory Epithelial Cells Convert Inactive Vitamin D to Its Active Form: Potential Effects on Host Defense
J. Immunol., 181 (2008), pp. 7090-7099, 10.4049/jimmunol.181.10.7090
CrossRefView Record in ScopusGoogle Scholar
[35]
A. Rafique, L. Rejnmark, L. Heickendorff, H.J. Møller25(OH)D 3 and 1.25(OH) 2 D 3 inhibits TNF-α expression in human monocyte derived macrophages
PLoS One., 14 (2019), 10.1371/journal.pone.0215383
Google Scholar
[36]
O. Andrukhov, O. Andrukhova, U. Hulan, Y. Tang, H.P. Bantleon, X. Rausch-FanBoth 25-hydroxyvitamin-D3 and 1,25-dihydroxyvitamin- D3 reduces inflammatory response in human periodontal ligament cells
PLoS One, 9 (2014), p. e90301, 10.1371/journal.pone.0090301
CrossRefGoogle Scholar
[37]
A. Simonnet, M. Chetboun, J. Poissy, V. Raverdy, J. Noulette, A. Duhamel, J. Labreuche, D. Mathieu, F. Pattou, M. Jourdain, R. Caizzo, M. Caplan, N. Cousin, T. Duburcq, A. Durand, A. El kalioubie, R. Favory, B. Garcia, P. Girardie, J. Goutay, M. Houard, E. Jaillette, N. Kostuj, G. Ledoux, D. Mathieu, A.S. Moreau, C. Niles, S. Nseir, T. Onimus, E. Parmentier, S. Préau, L. Robriquet, A. Rouze, S. Six, H. VerkindtHigh Prevalence of Obesity in Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) Requiring Invasive Mechanical Ventilation
Obesity., 28 (2020), pp. 1195-1199, 10.1002/oby.22831
CrossRefView Record in ScopusGoogle Scholar
[38]
A.R. Martineau, N.G. ForouhiVitamin D for COVID-19: a case to answer?
Lancet. Diabetes Endocrinol. (2020), 10.1016/S2213-8587(20)30268-0
Google Scholar
[39]
R.P. HeaneyGuidelines for optimizing design and analysis of clinical studies of nutrient effects
Nutr. Rev., 72 (2014), pp. 48-54, 10.1111/nure.12090
CrossRefView Record in ScopusGoogle Scholar
[40]
W.B. Grant, B.J. Boucher, H.P. Bhattoa, H. LahoreWhy vitamin D clinical trials should be based on 25-hydroxyvitamin D concentrations
J. Steroid Biochem. Mol. Biol., 177 (2018), pp. 266-269, 10.1016/j.jsbmb.2017.08.009
ArticleDownload PDFView Record in ScopusGoogle Scholar
[41]
R. Bouillon, D. BikleVitamin D Metabolism Revised: Fall of Dogmas
J. Bone Miner. Res., 34 (2019), pp. 1985-1992, 10.1002/jbmr.3884
CrossRefView Record in ScopusGoogle Scholar
[42]
D.A. Jolliffe, C. Stefanidis, Z. Wang, N.Z. Kermani, V. Dimitrov, J.H. White, J.E. McDonough, W. Janssens, P. Pfeffer, C.J. Griffiths, A. Bush, Y. Guo, S. Christenson, I.M. Adcock, K.F. Chung, K.E. Thummel, A.R. MartineauVitamin D Metabolism is Dysregulated in Asthma and Chronic Obstructive Pulmonary Disease
Am. J. Respir. Crit. Care Med. (2020), 10.1164/rccm.201909-1867oc
Google Scholar
[43]
J.M. Quesada-Gomez, R. BouillonIs calcifediol better than cholecalciferol for vitamin D supplementation?
Osteoporos. Int., 29 (2018), pp. 1697-1711, 10.1007/s00198-018-4520-y
CrossRefView Record in ScopusGoogle Scholar
[44]
RECOVERY Collaborative Group, P. Horby, W.S. Lim, J.R. Emberson, M. Mafham, J.L. Bell, L. Linsell, N. Staplin, C. Brightling, A. Ustianowski, E. Elmahi, B. Prudon, C. Green, T. Felton, D. Chadwick, K. Rege, C. Fegan, L.C. Chappell, S.N. Faust, T. Jaki, K. Jeffery, A. Montgomery, K. Rowan, E. Juszczak, J.K. Baillie, R. Haynes, M.J. LandrayDexamethasone in Hospitalized Patients with Covid-19 - Preliminary Report
N. Engl. J. Med. (2020), 10.1056/NEJMoa2021436
Google Scholar
[45]
Corticosteroids (including dexamethasone). NIH website. Updated July 17, 2020. Accessed August, 2020. Corticosteroids | Coronavirus Disease COVID-19.
Google Scholar
