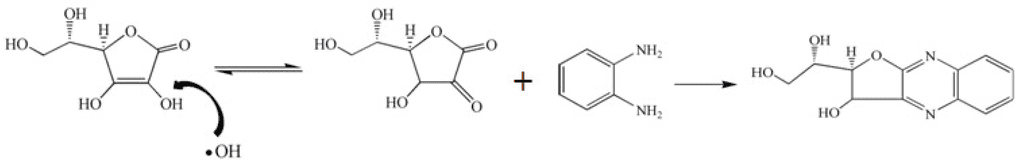
@haidut it's not just the contamination Ray seems to talk about, but the chemical reaction process? You'd have to source the Cevitamic acid not ascorbic acid, but Ray seems to suggest no one does the original method.
What other than contamination could be the issue? Peat's quotes from earlier in the thread talks about contamination only. What does the flawed manufacturing process do other than add contamination? Does it change the molecule somehow?
"...The same people who reacted to it often reacted similarly to riboflavin and rutin, which were also made from cornstarch by oxidation. I ascribed the reaction to some industrial contaminant that they had in common, possibly the heavy metals introduced with the sulfuric acid. The heavy metal contamination of synthetic ascorbate is so great that one 500 mg tablet dissolved in a liter of water produces free radicals at a rate that would require a killing dose of x-rays to equal. The only clean and safe vitamin C now available is that in fresh fruits, meats, etc. The commercial stuff is seriously dangerous."