Such_Saturation
Member
- Joined
- Nov 26, 2013
- Messages
- 7,370
I dunno man... long eyeball seems to me like a fitting feature of a vitamin A and light-deficient morphogenesis.I'm a midwesterner.
I would think—based on the tentative model—that less amounts of the more refractive methylimidazalone would be formed on the euglycemic state. This should decrease the refractive index of the lens after a few weeks—or however long it takes to form a new lens. I would also think the velocity of the change would depend on where exactly the rhodopsin is located relative to the focal point, and there being less change in those concomitantly wearing glasses. If the retina remodels faster than the lens, can keep‐up with the varying refractive index, you could perhaps expect less noticeable change.
Raising methylglyoxal to increase the refractive index might could be a thing to try. The two best ways for doing this, in my opinion, are by using a glyoxylase I inhibitor and/or L-threonine. Paul Thornally has written extensively on the glyoxylase system, and has published an excellent review. It basically consists of two enzymes—glyoxylase I and glyoxylase II—which transform endogenous methylglyoxal into lactate in a step-wise fashion, in series. The first step is methylglyoxal spontaneously associating with the thiol of glutathione: this new complex is the substrate for glyoxlase I, which simply isomerizes this and letting glyoxylase II do the rest—releasing lactate and regenerating glutathione. Inhibiting this enzyme leads to a higher steady‐state methylglyoxal level. The best inhibitors that I've seen were β-lapachone and lapachol, both found in pau d'arco. These molecules have the lowest ki values (~5·μM), and are both small and soluble—making them better options than the others. Curcumin—for instance—inhibits in vitro isolated glyoxylase I to a similar degree, but can barely penetrate the cell; in fact, it's barely absorbed at all. All the pharmacokinetic studies measure essentially zero blood levels after consuming even gram amounts.
But probably just as good for raising methylglyoxal is L-threonine. This is the only amino acid which becomes methylglyoxal directly in high amounts—a process process even mentioned by Vander Jagt in an article cited above.
"Methylglyoxal can also be made from aminoacetone during the catabolism of L-threonine (6)." —Vander Jagt
He cited (6) an article published in 1959, but there are better ones out there. The article he cites isn't experimental proof, but I've read articles that do actually prove this.
And it's easy to see why this should happen: After decarboxylation, threonine becomes aminoacetone; this is then acted-upon by monoamine oxidase forming methylglyoxal:
[pic]
The best way (and safest way) for raising methylglyoxal levels, in my opinion, is by consuming pau d'arco tea—in a french press, with coffee—while taking anywhere between 500 – 3,000 milligrams of L-threonine per day. Pau d'arco is actually an inner tree bark, like ceylon cinnamon, and tastes somewhat like cinnamon.
Thornalley, Paul J. "The glyoxalase system in health and disease." Molecular aspects of medicine (1993)
Thornalley Paul J. "Inhibitors of glyoxalase I: design, synthesis, inhibitory characteristics and biological evaluation." Biochemical Society transactions (1993)
Vander Jagt, D. L. "Reduction of trioses by NADPH-dependent aldo-keto reductases. Aldose reductase, methylglyoxal, and diabetic complications." Journal of Biological Chemistry (1992)
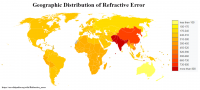
Well, check‐out the prevalence of refractive change in Asia:
View attachment 7394click to embiggen
I used to think that they wore glasses just to look smart, but.. . . ..but have you seen the List of Chinese inventions on Wikipædia? Not bad, if I may say so myself. The Japanese and Chinese could very well deserve the stereotype.
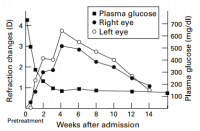
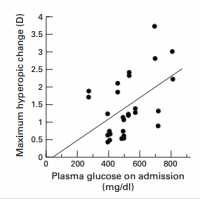
I think that it must be false, or misleading, in most cases. Hyperglycemia can cause refractive changes in minutes, and aldose reductase inhibitors prevent this from happening. This proves that glycolytic metabolism downstream of aldose reductase can cause refractive changes, and also deftly refutes the osmotic theories since sorbital or triose don't have any higher osmotic potential than glucose itself—or sodium for that matter. Of course, a change in the eyeballs's shape—or the position of the retina—would very likely produce changes in light perception (unless two parameters change simultaneously, thereby off‐setting eachother), but the associations between blood glucose levels are undeniable. This isn't the only way to produce myopia/hyperopia, but I think that's its the primary factor in the majority of cases. For some reason, this idea isn't popular; this could perhaps be the result of politics, or that most people prefer a simpler explanation—or a mix of the two.
You will! Hang in there. We need to read more about rhodopsin, microtubules, and cone cells. . . but not to discount the role of the refractive index of the lens itself—changed through a post‐translational modification or the lens protein crystallin, thereby producing an nonconventional amino acid higher refractive index.
I am willing to bet that we could find scientific articles describing how the morphology and growth rate of retinal cone cells change on exposure to light. These cells need to occupy the space of the focal point of the lens, and play a role in all of this.
Is this Ray Peat talking about the "glycation" of crystallin? I think this is a misleading term, as its too close to "glycosylation;" this almost implies that glucose itself is adding to the protein. This is not the case. Certain trioses are reacting with arginine side‐chains in a cyclicization reaction, in a manner which can probably be best described as a post‐translational modification. Thrornally has shown just recently that that methylglyoxal can induce or suppress DNA→mRNA transcription in this manner—by modifying a transcription factor in cell's nucleus (mSin3A).
And 'glycation' is also misleading for the obvious reason that methylglyoxal can be produced from either glycerol of threonine.
Yao, Dachun. "High glucose increases angiopoietin-2 transcription in microvascular endothelial cells through methylglyoxal modification of mSin3A." Journal of Biological Chemistry (2007)
Wang, Tina. "Exploring post-translational arginine modification using chemically synthesized methylglyoxal hydroimidazolones." Journal of the American Chemical Society (2012)
I had realized a few days ago that I had to look into this, and that glucose must be involved–somehow. When reading about it, it because clear what was going on. This led directly into a more familiar territory form me: the methylglyoxal–arginine adducts, what I find most interesting.
You'd expect this to occur, but γ-crystallin has only eight cysteines; compare that to the twenty arginines. The methylglyoxal–thiol bond is a bit more labile that the condenstation of methylglyoxal and arginine, which forms a species so resilient that it can be detected in urine. Thiol–acyl bonds, as is acetyl–CoA, are routinely formed and cleaved by low-energy enzymes within the body during (fatty acid metabolism). The carbon–sulfur bond isn't one of the strongest, and I'd assume any methylglyoxal–thiol bonds to be transient (as in the case of methylglyoxal–glutathione); sulfur binds most strongly to mercury.
Glutathione is needed to turn methylglyoxal into lactic acid, and can perhaps be considered a cofactor. Glutathione is a prerequisite for the methylgyloxal→lactic acid conversion through the glyoxylase system. High methylglyoxal leads to less-active glutathione as it's occupied by such, on the thiol group. This hasn't been overlooked in attempting to explain how methylglyoxal reverses cancer; it has been speculated that this occupation of glutathione is the primary reason. While it's true that gluationine is needed for mitosis, I think a bit differently: I think that methylglyoxal works mostly by adding to a specific arginine side-chain in NADH-dependent glycolytic enzymes, which all have an arginine which bonds the phosphodiester of it's cofactor—NADH. Arginine is always found at this location in such enzymes in a highly‐intuitive interaction: The fully-positive or ∂-positive charges of arginine—found on the γ-nitrogens—interact noncovalently with the negative oxygen atoms of the phosphodiester—like the well-known 'salt bridge' between glutamate and lysine, for instance. These NADH-dependent enzymes simply cannot bind the cofactor without this arginine, and point-mutational analysis fully confirm this. This would explain why glyolytic rates drop quickly with 3‐bromopyruvate or methylglyoxal: They work on glycolytic enzymes directly by abrogating their ability to bind NADH—their cofactor—by permanently‐modifying a key arginine residue while it's being assembled in the ribosome . . . or shortly thereafter.
That's a very interesting observation, and an interesting graph. Growth hormone does raise insulin, right? leading to lower blood glucose and methylglyoxal—leading then further to a lower percentage of arginine transformed into methylimidazolone resulting in a negative shift of the lens' refractive index.
This is interesting. I know that glutamate and GTP both powerfully influence the polymerization rates of tubulin in vitro, and had a thought that dopamine was perhaps involved in this. If you look at colchicine, you'll realize it has multiple methoxy groups. You may also realize that over 95% of molecules classified as "microtubule inhibitors" have methoxy groups—you can find dozens of examples of this. Dopamine can be methylated on one of the catechol oxygens, leading to methoxytyramine—or methyldopamine (synonymous). The enzyme catechol‐O‐methyltransferase is responsible for doing this, and the low‐activity methionine‐encoding allele of the gene is often correlated with higher IQ. There are dozens of studies on catechol‐O‐methyltransferase and IQ, and methoxytyramine has been implicated in Parkinson's dyskinesia (caused by ingesting too much L-DOPA.)
I don't find it hard to believe that the ratio of dopamine to methyldopamine could influence microtubules in vivo, since the methoxy groups seems almost a prerequisite for a powerful microtubule polymerization inhibitor/destabilizer (see colchicine, methoxyestradiol, and many more.)
Yeah. You'd think that polyunsaturated fatty acids would activate the Randall Cycle, likely through PPARγ upregulating the fatty acid synthase and the β-oxidation enzymes. Too many fatty acids of the wrong type certainly seem capable of throwing someone into 'diabetes mode.' Anything that effects glucose metabolism—or modulates the glyoxylase system—would be expected to modify the refractive index of crystallin. The higher prevalence of myopia among adolescents can perhaps be explained by the action of growth hormone, through raising insulin (Diokene, 2017); and the hyperopia seen in diabetes can certainly by caused by hyperglycemia (Eva, 1982).