Travis
Member
- Joined
- Jul 14, 2016
- Messages
- 3,189
Pellagra is fairly well understood, but there is still some persistent mythology surrounding the etiology. Everyone knows that niacin is the characteristic deficiency, but few are aware of the central importance that leucine plays in this. The somewhat low tryptophan and niacin content of corn is stressed, with little attention given to the main causative agent—if any.
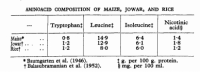
Sorgham causes pellagra, yet has a fair amount of tryptophan and and free niacin. The most salient commonality between sorgham and corn is their extraordinarily high leucine concentrations, as expressed as the ratio to total protein.
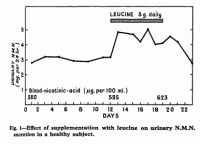
What is unique about leucine is its ability to increase the elimination of nicotinic acid (B₃). In a study on adult males over fifty years ago, Gopalan showed the extent of this effect (using only five grams of leucine):
This is a common observation, seen in animals of all types. Isoleucine or valine doesn't do this; this effect is particular to leucine.
Nixtamalization is process where the corn is ground, mixed with water, mixed with alkali, and then strained. This represents a separation procedure, and is known to ameliorate the pellagric effect of corn. The change in amino acid composition has been studied, with significant reductions in the leucine content observed.
This is best explained by the fact that the increased pH causes a precipitation of the protein zein, the alcohol‐soluble fraction. This is then discarded with the water, along with much of the leucine content. Nixtamalization represents a balancing of the amino acid profile. The idea that this process serves to 'liberate niacin' is naïve, considering the fact that sorghum—and even leucine itself—has been shown to cause pellagra. Reading mythological narratives about 'bound niacin' leaves the reader befuddled; it is never explained what it's bound to. Accepting this is akin to accepting the notion that corn has a peculiar and ill‐defined ability to bind niacin, an effect reversed by alkalinity in vitro but not by alkalinity in vivo (duodenal). Moreover, the mythological resilient niacin–protein bond is necessarily unaffected by the hydrochloric acid, the pepsin, the trypsin, and the twelve hours of bacterial action in the digestive tract.
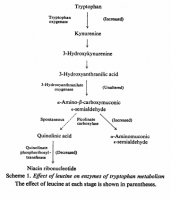
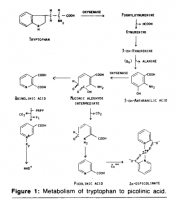
But this explanation fails, for example, to explain why leucine increases tryptophan oxygenase activity; the entire kyneurenine pathway is increased, and shifted. Relatively small changes in leucine divert the kyneurenine pathway from the niacin arm to the picolinate arm:
The data does not suggest a simple inhibition of one enzyme; the data suggests a purposeful adaption to excessive leucine, forming picolinate at the expense of niacin.
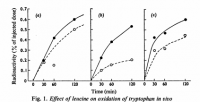
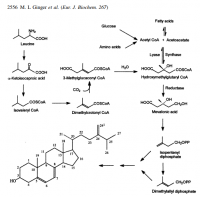
Excessive amino acids are generally decarboxylated first, then perhaps deanimated. First glance at leucine gives hints about what it could be used for: Leucine's carbon skeleton—sans carbon dioxide—is essentially a saturated isoprene, inviting the speculation that it could be used in cholesterol synthesis. This speculation turns out to be correct, and ¹⁴C‐leucine has been shown to be incorporated into sterols in three Leishmania species—and even in rats:
In both protazoa and mammals, leucine can serve as a substrate for steroid synthesis—a fact of central importance to lecucine‐induced pellagra.
Picolinate is no stranger to biology, and perhaps its most particular feature is its ability to chelate zinc. Even commercially, zinc is commonly sold as a picolinate chelate. This is a high‐affinity bond which has biological importance. Picolinate supplementation increases the absorption of dietary zinc, and it wouldn't be a stretch to imagine endogenous picolinate increases the absorption of zinc. This turns out to be true, as was proven by G.W. Evans:
This is another fact to keep in mind—a necessary step in explaining why the body does what it has been shown to do, in response to excess leucine.
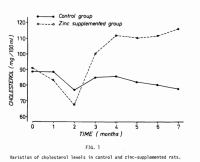
Zinc increases cholesterol biosynthesis. This is another consistent finding, which is observed in humans with zinc supplementation.
This is also observed in rats, and the inclusion of zinc to their diet predictable lead to hypercholesterolemia.
'However, several studies have shown a role for zinc in determining serum cholesterol and triglyceride levels: a zinc-deficient diet induces hypotriglyceridemia in rats and hypocholesterolemia in rats and pigs; zinc supplemented swines had higher cholesterol levels than their unsupplemented counterparts during the first 4 weeks of treatment; Lei using a moderately copper-deficient diet supplemented with zinc obtained a hypercholesterolemia in the rat.' ―Katya-Katya
The regulatory cascade which orchestrates cholesterol synthesis is zinc‐dependent. The enzyme required in the second canonical stage needs zinc ions to function, a fact which can explain how dietary zinc can effect cholesterol levels so powerfully.
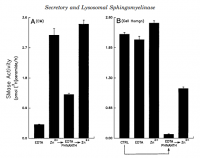
Besides site‐2‐protease, other zinc metalloproteases are involved in lipid synthesis. A study conducted in 1998 shows the zinc dependency of sphingomyelinase directly.
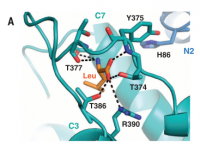
The enzyme responsible for the second, and indispensible, step in canonical cholesterol regulation has been characterized. It has been found to contain a considerable amount of zinc, bound tightly, coordinated by histidine (of course):
Radioactive ¹⁴C‐leucine has been shown to be incorporated into the cholesterol skeleton by mammals, including mice; picolinate has been shown to chelate zinc, and so has tryptophan (ostensibly via picolinate); zinc has unambiguously been shown to increase cholesterol synthesis; what follows from this is an explanation why dietary leucine increases the production of picolinate at the expense of niacin: It purposefully does so as a device to increase the absorption of the obligatory dietary zinc for cholesterol synthesis, the most efficient way for the body to deal with excessive leucine.
Sorgham causes pellagra, yet has a fair amount of tryptophan and and free niacin. The most salient commonality between sorgham and corn is their extraordinarily high leucine concentrations, as expressed as the ratio to total protein.
'A comparison of the chemical composition of rice, jowar, and maize shows that the nicotinic acid content of jowar is nearly similar to that of rice. The tryptophan content of jowar varies widely, certain strains having nearly as high a content of tryptophan as rice, while certain others have somewhat lower values. Both jowar and maize have, however, one common feature with regard to their amino acid composition: a high content of leucine.' ―Gopalan
'Experimental pellagra was produced in six adult monkeys by feeding diets containing jowar. Clinical and pathological features of this syndrome were indistinguishable from those seen in a group which developed the deficiency on maize diet.' ―Gopalan
What is unique about leucine is its ability to increase the elimination of nicotinic acid (B₃). In a study on adult males over fifty years ago, Gopalan showed the extent of this effect (using only five grams of leucine):
'The rise in N.M.N. [N‐methyl nicotinic acid] excretion brought about by 5 grams L‐leucine daily represented an increase of nearly 50% over the basal level.'―Gopalan
'The increase in N.M.N. excretion brought about by supplementation with 5 grams of L‐leucine daily would roughly correspond to the metabolism of an additional 3–5 milligram nicotinic acid daily.' ―Gopalan
This is a common observation, seen in animals of all types. Isoleucine or valine doesn't do this; this effect is particular to leucine.
Gopalan, C. "Leucine and pellagra." The Lancet (1960)
Gopalan, C. "Leucine and pellagra." Nutrition reviews (1968)
Gopalan, C. "Leucine and pellagra." Nutrition reviews (1968)
Nixtamalization is process where the corn is ground, mixed with water, mixed with alkali, and then strained. This represents a separation procedure, and is known to ameliorate the pellagric effect of corn. The change in amino acid composition has been studied, with significant reductions in the leucine content observed.
'From these figures the average amino acid changes were then calculated. There was a 21.0% decrease for leucine, 18.7% for arginine, 12.5%, for cystine, 11.7% for histidine, and 5.3% for lysine. Of the nonessential amino acids, changes were found in proline, serine, and glutamic acid, amounting respectively to a decrease of 8.7, 7.1, and 6.3%.' —Bressani
'On the other hand, an average of 21% of leucine was lost in the process, a change which should improve the biological value of tortilla protein by partially correcting the isoleucine to leucine disproportion.' —Bressani
This is best explained by the fact that the increased pH causes a precipitation of the protein zein, the alcohol‐soluble fraction. This is then discarded with the water, along with much of the leucine content. Nixtamalization represents a balancing of the amino acid profile. The idea that this process serves to 'liberate niacin' is naïve, considering the fact that sorghum—and even leucine itself—has been shown to cause pellagra. Reading mythological narratives about 'bound niacin' leaves the reader befuddled; it is never explained what it's bound to. Accepting this is akin to accepting the notion that corn has a peculiar and ill‐defined ability to bind niacin, an effect reversed by alkalinity in vitro but not by alkalinity in vivo (duodenal). Moreover, the mythological resilient niacin–protein bond is necessarily unaffected by the hydrochloric acid, the pepsin, the trypsin, and the twelve hours of bacterial action in the digestive tract.
Kodicek, E. "The availability to pigs of nicotinic acid in tortilla baked from maize treated with lime-water." British Journal of Nutrition (1959)
Bressani, Ricardo. "Lime-heat effects on corn nutrients, effect of lime treatment on in vitro availability of essential amino acids and solubility of protein fractions in corn." Journal of Agricultural and Food chemistry (1958)
Why exactly would leucine—of all things—increase the elimination of niacin? So far, I have yet to see an acceptable answer for this. All the articles I've read on this topic leave the question open; there has been little attempt to explain why this should be. Naturally, there are those who think responses like this happen by accident; the explanation that 'leucine inhibits quinolinate phosphoribosyl transferase' (wikipedia.org) is an acceptable explanation for those people. Bressani, Ricardo. "Lime-heat effects on corn nutrients, effect of lime treatment on in vitro availability of essential amino acids and solubility of protein fractions in corn." Journal of Agricultural and Food chemistry (1958)
'Nature does not indulge in luxuries and if there is such a widely spread and highly active enzyme, it must have something very important to do...' ―Albert Szent‐Györgyi
But this explanation fails, for example, to explain why leucine increases tryptophan oxygenase activity; the entire kyneurenine pathway is increased, and shifted. Relatively small changes in leucine divert the kyneurenine pathway from the niacin arm to the picolinate arm:
'To understand the biochemical basis of leucineinduced changes in tryptophan-niacin metabolites, the effects of dietary leucine on the enzymes involved in tryptophan-niacin metabolism were investigated. The enzymes studied were tryptophan oxygenase, 3-hydroxyanthranilate oxygenase, picolinate carboxylase, quinolinate phosphoribosyltransferase and nicotinate phosphoribosyltransferase.' ―Narasinga
'A decreased activity of quinolinate phosphoribosyltransferase may also partly explain the increased quinolinic acid excretion observed in the presence of excess of leucine in the diet (Raghuramulu et al., 1965a; Belavady et al., 1963). The fact that leucine in the presence of added niacin decreased the enzyme activity without affecting quinolinic acid excretion (Raghuramulu et al., 1965a) suggests other mechanisms contributing to increased quinolinic acid excretion.' ―Narasinga
The data does not suggest a simple inhibition of one enzyme; the data suggests a purposeful adaption to excessive leucine, forming picolinate at the expense of niacin.
Excessive amino acids are generally decarboxylated first, then perhaps deanimated. First glance at leucine gives hints about what it could be used for: Leucine's carbon skeleton—sans carbon dioxide—is essentially a saturated isoprene, inviting the speculation that it could be used in cholesterol synthesis. This speculation turns out to be correct, and ¹⁴C‐leucine has been shown to be incorporated into sterols in three Leishmania species—and even in rats:
'The results presented here demonstrate that leucine acts as a major precursor to sterols in several Leishmania species and also in the closely related organism E. monterogeii. Some Leishmania species derived considerably more of their sterol from a leucine source of carbon than did others (Table 3) but there was no apparent correlation between the extent of leucine utilization for sterol production and the origin of the organism as an Old World or New World species.' ―Ginger
'The conversion of leucine to cholesterol in muscle tissue was about 200 times that of acetate and about 25 times that of mevalonate. Utilisation of leucine for cholesterol synthesis was greater than that of acetate and of mevalonate even in intestine and kidney, whereas in liver homogenate acetate and mevalonate were better precursors than leucine.' ―Miettinen
In both protazoa and mammals, leucine can serve as a substrate for steroid synthesis—a fact of central importance to lecucine‐induced pellagra.
Szent-Györgyi, Albert. "The living state and cancer." Proceedings of the National Academy of Sciences (1977)
Rao, BS Narasinga. "Effect of leucine on enzymes of the tryptophan–niacin metabolic pathway in rat liver and kidney." Biochemical Journal (1973)
Ginger, Michael L. "The biosynthetic incorporation of the intact leucine skeleton into sterol by the trypanosomatid Leishmania mexicana." Journal of Biological Chemistry (2001)
Miettinen, T. A. "Comparison of leucine with mevalonate and acetate as a precursor of tissue and serum cholesterol in the rat." Annales medicinae experimentalis et biologiae Fenniae (1971)
Ginger, Michael L. "Utilization of leucine and acetate as carbon sources for sterol and fatty acid biosynthesis by Old and New World Leishmania species, Endotrypanum monterogeii and Trypanosoma cruzi." The FEBS Journal (2000)
Rao, BS Narasinga. "Effect of leucine on enzymes of the tryptophan–niacin metabolic pathway in rat liver and kidney." Biochemical Journal (1973)
Ginger, Michael L. "The biosynthetic incorporation of the intact leucine skeleton into sterol by the trypanosomatid Leishmania mexicana." Journal of Biological Chemistry (2001)
Miettinen, T. A. "Comparison of leucine with mevalonate and acetate as a precursor of tissue and serum cholesterol in the rat." Annales medicinae experimentalis et biologiae Fenniae (1971)
Ginger, Michael L. "Utilization of leucine and acetate as carbon sources for sterol and fatty acid biosynthesis by Old and New World Leishmania species, Endotrypanum monterogeii and Trypanosoma cruzi." The FEBS Journal (2000)
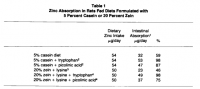
Picolinate is no stranger to biology, and perhaps its most particular feature is its ability to chelate zinc. Even commercially, zinc is commonly sold as a picolinate chelate. This is a high‐affinity bond which has biological importance. Picolinate supplementation increases the absorption of dietary zinc, and it wouldn't be a stretch to imagine endogenous picolinate increases the absorption of zinc. This turns out to be true, as was proven by G.W. Evans:
'Those investigators noted that the metabolism of tryptophan was impaired in children who were affected with acrodermatitis enteropathica and suggested that the genetic defect in this disease involved an enzyme in the pathway from tryptophan to nicotinic acid. We believed that this malady resulted from an impaired production of an essential zinc-binding ligand. We thus investigated the possibility that a tryptophan metabolite might be the long sought after zinc binding ligand.' ―Evans
'Rats fed a zein-based diet (20 percent protein) supplemented with lysine but not with tryptophan absorbed only 59 percent of the daily dietary zinc intake. Rats fed the zein diet supplemented with both lysine and tryptophan absorbed 98 percent of the dietary zinc intake, and rats fed the zein diet supplemented with lysine and picolinic acid absorbed 75 percent of the dietary zinc intake (Table 1).' ―Evans
'These observations establish the fact that zinc absorption is affected by levels of both dietary tryptophan and pyridoxine. Furthermore, the results demonstrate that supplemental picolinic acid ameliorates the impaired zinc absorption caused by a deficiency of either tryptophan or pyridoxine. Because picolinic acid is a metabolic product of tryptophan and depends upon pyridoxine for its production, these results provide strong evidence that endogenous picolinic acid is essential for normal zinc absorption.' ―Evans
'The studies reviewed suggest that zinc absorption proceeds as follows: in the exocrine cells of the pancreas the metabolism of tryptophan produces picolinic acid which is secreted into the lumen of the intestine. In the lumen, picolinic acid coordinates with Zn²⁺ to form a complex which facilitates the passage of Zn²⁺ through the luminal membrane, across the absorptive cell and through the basolateral membrane of the cell.' ―Evans
This is another fact to keep in mind—a necessary step in explaining why the body does what it has been shown to do, in response to excess leucine.
Evans, G. W. "Normal and abnormal zinc absorption in man and animals: the tryptophan connection." Nutrition reviews (1980)
Seal, Christopher J. "Effect of dietary picolinic acid on the metabolism of exogenous and endogenous zinc in the rat." The Journal of nutrition (1985)
Seal, Christopher J. "Effect of dietary picolinic acid on the metabolism of exogenous and endogenous zinc in the rat." The Journal of nutrition (1985)
Zinc increases cholesterol biosynthesis. This is another consistent finding, which is observed in humans with zinc supplementation.
'On day 43, there was a significant elevation in the serum total cholesterol for subjects in the zinc-supplemented group (191.44 mg/dL) compared with the control group (151.42 mg/dL). No significant changes were noted in the serum total cholesterol in the control group from day 0 (146.61 mg/dL) to day 43 (151.42 mg/dL).' ―Roozbeh
This is also observed in rats, and the inclusion of zinc to their diet predictable lead to hypercholesterolemia.
'However, several studies have shown a role for zinc in determining serum cholesterol and triglyceride levels: a zinc-deficient diet induces hypotriglyceridemia in rats and hypocholesterolemia in rats and pigs; zinc supplemented swines had higher cholesterol levels than their unsupplemented counterparts during the first 4 weeks of treatment; Lei using a moderately copper-deficient diet supplemented with zinc obtained a hypercholesterolemia in the rat.' ―Katya-Katya
'At the end of the experiment, the zinc-supplemented rats had a 50% higher cholesterol concentration than the control rats.' ―Katya-Katya
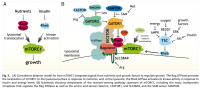
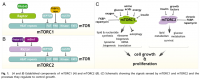
The regulatory cascade which orchestrates cholesterol synthesis is zinc‐dependent. The enzyme required in the second canonical stage needs zinc ions to function, a fact which can explain how dietary zinc can effect cholesterol levels so powerfully.
'Site-2-protease (S2P), a member of the family of zinc metalloproteases, catalyzes hydrolysis of the second proteolytic reaction on SREBP [sterol regulatory element binding protein], which releases a soluble transcription factor (the amino-terminal fragment of SREBP). The soluble transcription factor enters the nucleus, where it binds to direct repeat sterol response elements in promotor regions, activating transcription of sterol-regulated cholesterogenic genes. Genes under this regulatory control include, at least, LDL receptor, HMG-CoA synthase, HMG-CoA reductase, farnesyl diphosphate synthase, squalene synthase, lanosterol synthase, and lanosterol 14α-methyl demethylase cytochrome P450. Activation of these genes restores intracellular cholesterol levels.' ―Risley
''The first RIP system to be molecularly characterized was the cleavage of the membrane-anchored transcription factor SREBP (sterol regulatory element–binding protein) by a metalloprotease known as site-2 protease (S2P). This cleavage releases a transcription factor, which translocates into the nucleus of the cell and activates genes involved in synthesis and uptake of cholesterol and fatty acids.' ―Feng
Besides site‐2‐protease, other zinc metalloproteases are involved in lipid synthesis. A study conducted in 1998 shows the zinc dependency of sphingomyelinase directly.
'We then tested this purified L-SMase for zinc dependence and found that its enzymatic activity was increased 4.7-fold in the presence of Zn²⁺.' ―Schissel
'the data in this report strongly support the conclusion that this enzyme is, indeed, a zinc‐activated enzyme.' ―Schissel
The enzyme responsible for the second, and indispensible, step in canonical cholesterol regulation has been characterized. It has been found to contain a considerable amount of zinc, bound tightly, coordinated by histidine (of course):
'S2P has six transmembrane segments, TM1 through TM6. The catalytic zinc atom is located ~14 Å from the lipid membrane surface. Zinc is coordinated by three amino acids, His⁵⁴ and His⁵⁸ in TM2, and Asp¹⁴⁸ in TM3, which are highly conserved in all S2P proteins.' ―Feng
'As previously hypothesized, the catalytic zinc atom is coordinated by three amino acids that are invariant among all S2P family proteins: His⁵⁴ and His⁵⁸ from helix a2 and Asp¹⁴⁸ from the N‐terminal end of helix [...]. The zinc atom is located ~14 Å below the lipid membrane surface from the cytosolic side.' ―Feng
'Quantitative element analysis by inductively coupled plasma emission spectrometry revealed that, compared with the WT protein, the zinc content was 8.2, 44.2, 5.1, and 15.1% for the mutant proteins H54A, E55A, H58A, and D148A, respectively.' ―Feng
'As previously hypothesized, the catalytic zinc atom is coordinated by three amino acids that are invariant among all S2P family proteins: His⁵⁴ and His⁵⁸ from helix a2 and Asp¹⁴⁸ from the N‐terminal end of helix [...]. The zinc atom is located ~14 Å below the lipid membrane surface from the cytosolic side.' ―Feng
'Quantitative element analysis by inductively coupled plasma emission spectrometry revealed that, compared with the WT protein, the zinc content was 8.2, 44.2, 5.1, and 15.1% for the mutant proteins H54A, E55A, H58A, and D148A, respectively.' ―Feng
Radioactive ¹⁴C‐leucine has been shown to be incorporated into the cholesterol skeleton by mammals, including mice; picolinate has been shown to chelate zinc, and so has tryptophan (ostensibly via picolinate); zinc has unambiguously been shown to increase cholesterol synthesis; what follows from this is an explanation why dietary leucine increases the production of picolinate at the expense of niacin: It purposefully does so as a device to increase the absorption of the obligatory dietary zinc for cholesterol synthesis, the most efficient way for the body to deal with excessive leucine.
Risley, John M. "Cholesterol biosynthesis: Lanosterol to cholesterol." J. Chem. Educ (2002)
Feng, Liang. "Structure of a site-2 protease family intramembrane metalloprotease." Science (2007)
Katya-Katya, M. "The effect of zinc supplementation on plasma cholesterol levels." Nutrition research (1984)
Roozbeh, Jamshid. "Effect of zinc supplementation on triglyceride, cholesterol, LDL, and HDL levels in zinc-deficient hemodialysis patients." Renal failure (2009)
Schissel, Scott L. "The cellular trafficking and zinc dependence of secretory and lysosomal sphingomyelinase, two products of the acid sphingomyelinase gene." Journal of Biological Chemistry (1998)
Feng, Liang. "Structure of a site-2 protease family intramembrane metalloprotease." Science (2007)
Katya-Katya, M. "The effect of zinc supplementation on plasma cholesterol levels." Nutrition research (1984)
Roozbeh, Jamshid. "Effect of zinc supplementation on triglyceride, cholesterol, LDL, and HDL levels in zinc-deficient hemodialysis patients." Renal failure (2009)
Schissel, Scott L. "The cellular trafficking and zinc dependence of secretory and lysosomal sphingomyelinase, two products of the acid sphingomyelinase gene." Journal of Biological Chemistry (1998)
Last edited: