Mito
Member
- Joined
- Dec 10, 2016
- Messages
- 2,554
“Federal officials said the pill, known as Paxlovid, will be a vital tool in treating people who test positive, develop symptoms and are at higher risk of a bad outcome, though they warned that quantity might be limited due to the complex chemistry involved in making the pills.”
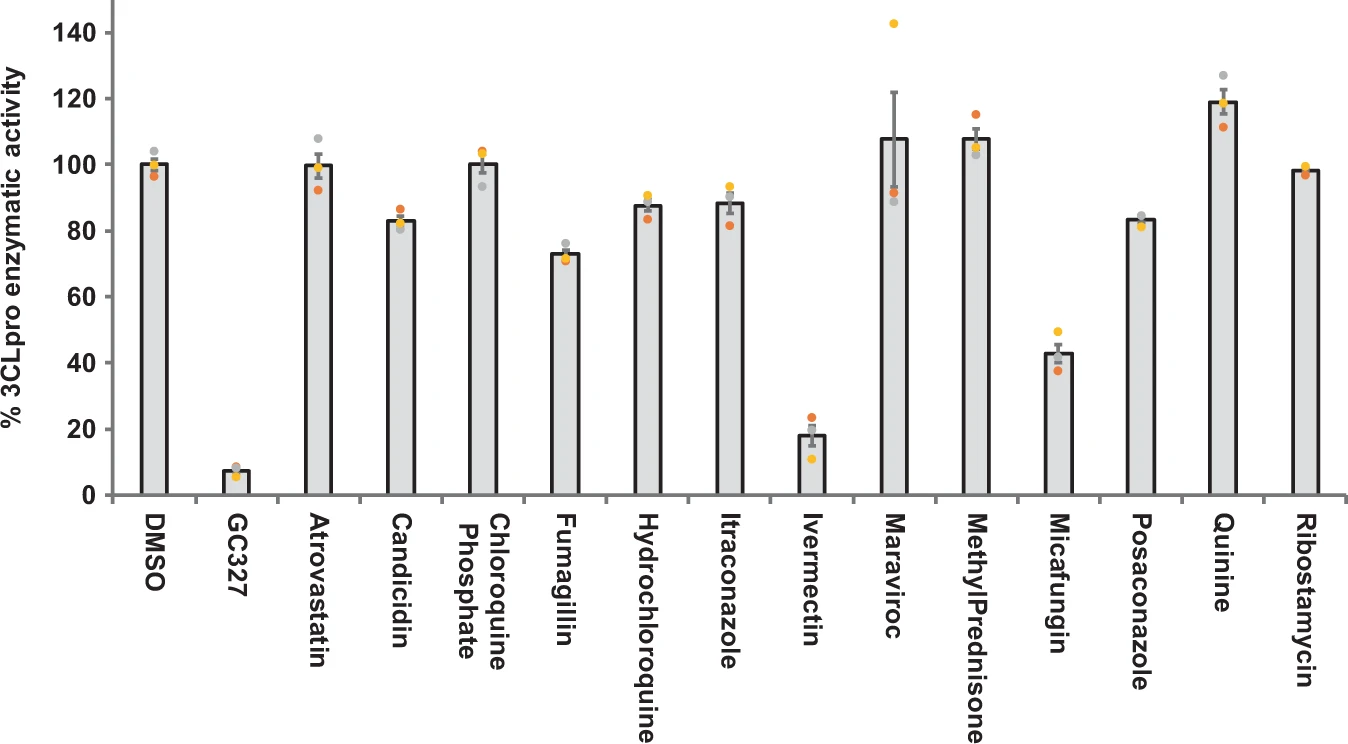
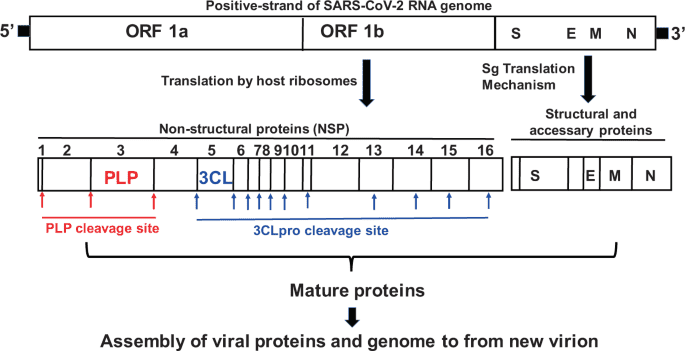
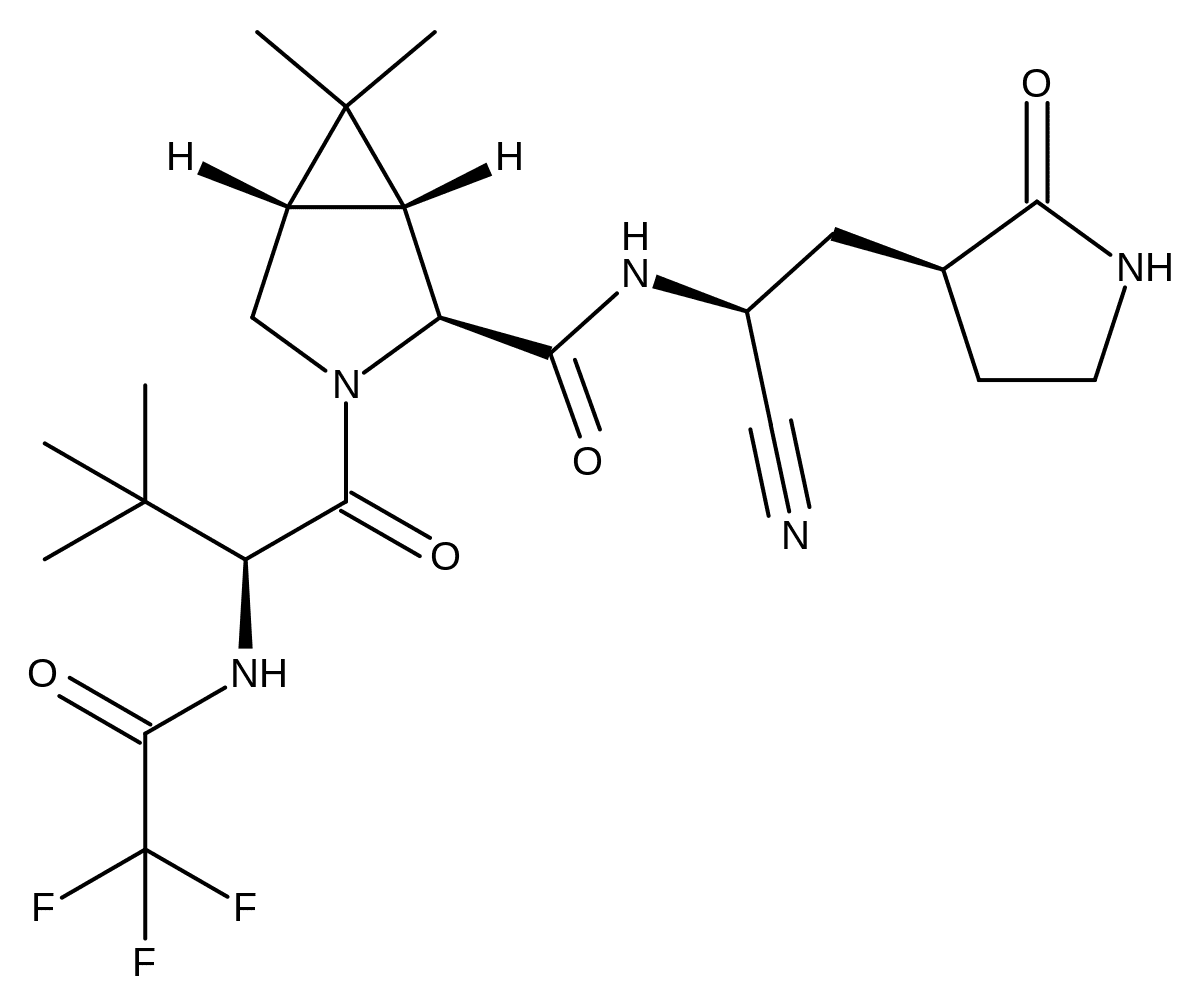
“Nirmatrelvir is an antiviral drug developed by Pfizer which acts as an orally active 3CL protease inhibitor.[2][3][4][5] In December 2021, the combination of nirmatrelvir with the CYP3A4 inhibitor ritonavir was given Emergency Use Authorization by the FDA for the treatment of COVID-19 in high-risk patients over the age of 12 under the brand name Paxlovid.[6][7]”
 en.wikipedia.org
en.wikipedia.org
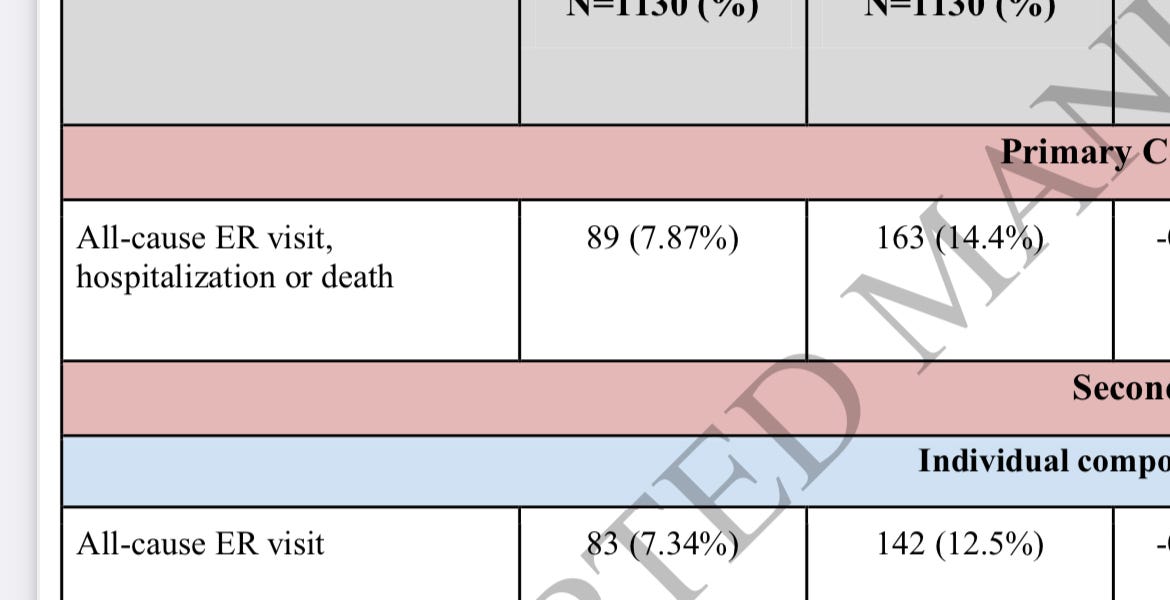
Pfizer oral COVID-19 pill gets U.S. authorization for at-home use
The FDA cleared use of Pfizer’s Paxlovid pill, the first Covid-19 drug that newly infected patients can now take at home to stay out of the hospital.
ground.news
“Nirmatrelvir is an antiviral drug developed by Pfizer which acts as an orally active 3CL protease inhibitor.[2][3][4][5] In December 2021, the combination of nirmatrelvir with the CYP3A4 inhibitor ritonavir was given Emergency Use Authorization by the FDA for the treatment of COVID-19 in high-risk patients over the age of 12 under the brand name Paxlovid.[6][7]”