David PS
Member
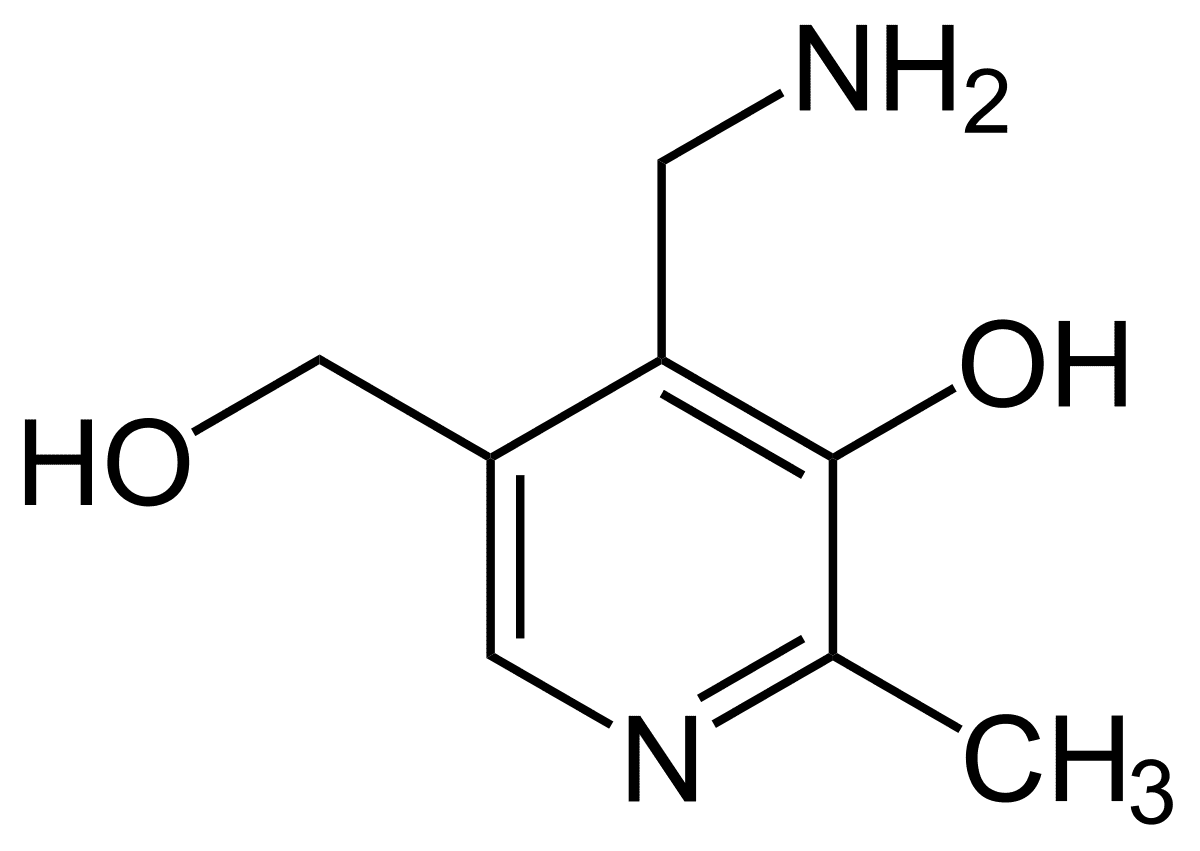
David's Sinclair has spoken about a new NMN pharmaceutical under development (MIB-626). Incidently, David is a co-founder of Metro International Biotech
 www.nmn.com
www.nmn.com
In addition, David is a named inventor on the US patent application assigned to Metro.
The two relevant documents describing the FDA's justification are at this deep link to the relevant page:
 www.regulations.gov
From the end of the second and most recent document (supplemental response letter)
www.regulations.gov
From the end of the second and most recent document (supplemental response letter)

Metro Biotech Publishes MIB-626 NMN Human Data
A study published by a research team from Metro International Biotech and Harvard Medical School shows that a pharmaceutical-grade preparation of NMN known as MIB-626 increases blood levels of NAD+ in one week in middle-aged and older adults.
In addition, David is a named inventor on the US patent application assigned to Metro.
The two relevant documents describing the FDA's justification are at this deep link to the relevant page:
Regulations.gov
3. Conclusions
After evidence that is irrelevant under section 201(ff)(3)(B) for the reasons discussed above has been eliminated, two possible dates of first marketing for NMN are left:
February 16, 2021 – First date on which another notifier could have marketed its NMNcontaining dietary supplement lawfully after submitting an NDI notification to FDA on December 3, 2020; however, it is unclear whether NMN was actually marketed on that date.
September 2021 – Month during which Kingdomway first marketed Doctor’s Best Instant Whey Protein Concentrate Plus NMN as a conventional food. Both of these dates are after the first date on which NMN was authorized for investigation as a new drug.24 Accordingly, we reaffirm our earlier conclusion that NMN is excluded from the dietary supplement definition under 21 U.S.C. § 321(ff)(3)(B)(ii) and may not be marketed as or in a dietary supplement. Today we are sending letters communicating this conclusion simultaneously to all firms that have submitted an NDI notification for NMN.:
Last edited: