Giraffe
Member
- Joined
- Jun 20, 2015
- Messages
- 3,730
Thalidomide was promoted as a sleeping pill and to help with morning sickness. Irreversible polyneuropathy was observed, and after the drug was linked to horrific birth defects it was banned.
It has since made a comeback. In 1998 Celgene launched thalidomide as a leprosy drug. The approval for leprosy was used as an entry point. Soon the drug was used off-label in blood cancers and AIDS. In 2006 it was approved for the treatment of multiple myeloma.
Thalidomide was investigated for many other diseases. A phase III trial for the use in Crohn's disease was discontinued in 2017.
This are the adverse effects from a Crohn's trial:

Long-term outcomes of thalidomide in refractory Crohn’s disease
Celgene is successfully keeping off competitors, and the price for the drug has sky-rocketed.
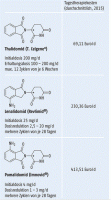
Next generation versions with supposedly less side effects are:
source
In 2017 Celgene agreed to pay $ 280 Million to resolve cancer fraud allegations.
Revlimid is predicted to be among the top 10 drugs by annual revenue in 2025.
It has since made a comeback. In 1998 Celgene launched thalidomide as a leprosy drug. The approval for leprosy was used as an entry point. Soon the drug was used off-label in blood cancers and AIDS. In 2006 it was approved for the treatment of multiple myeloma.
Thalidomide was investigated for many other diseases. A phase III trial for the use in Crohn's disease was discontinued in 2017.
This are the adverse effects from a Crohn's trial:
Long-term outcomes of thalidomide in refractory Crohn’s disease
Celgene is successfully keeping off competitors, and the price for the drug has sky-rocketed.
How A Drugmaker Gamed The System To Keep Generic Competition Away
How Celgene uses safety concerns to keep its $7 billion cancer drug on top
How Celgene uses safety concerns to keep its $7 billion cancer drug on top
Next generation versions with supposedly less side effects are:
- Revlimid (Lenalidomide)
- Imnovid (Pomalidomid)

source
In 2017 Celgene agreed to pay $ 280 Million to resolve cancer fraud allegations.
The payment is equivalent to about two weeks’ worth of sales of Revlimid, which generated $6.97 billion in revenue for Celgene last year, according to data compiled by Bloomberg. Celgene Settles Cancer Drug Whistleblower Suit
Revlimid is predicted to be among the top 10 drugs by annual revenue in 2025.

