@haidut
It seems mno2 can degrade bpa’s ,reduce glutathione in cancer cells and was combined with methylene blue inhibited tumor growth.
And mno2 was shown to reduce methylene blue and @haidut talked about methylene blue with ascorbic acid and turning it colorless and having some benefits.
This is above my pay grade, is there anything to this?
 www.nature.com
www.nature.com
The following reactions illustrate how acid-induced H2O2 promotes the degradation of MnO2 and the resultant generation of oxygen:
MnO2 shows high reactivity and specificity to acidic environments and the presence of H2O2, where it can produce oxygen continuously to overcome hypoxia in tumors42,43,44,45. After reacting with excessive H2O2 in an acidic tumor microenvironment, MnO2 nanosheets can be degraded to produce a large amount of oxygen, thus greatly improving the efficiency of PDT. In addition to increasing the oxygen content, MnO2 can also be used as a glutathione (GSH) inhibitor in vivo to increase PDT efficacy. Moreover, it can be used to make upconversion nanocomposites to stimulate PSs at various wavelengths to enhance the selectivity of PDT. In addition, Mn2+ produced by MnO2 is a strong T1 magnetic resonance (MR) contrast agent for tumor detection, which can also be used for in site tumor imaging46,47. At the same time, it is worth mentioning that, as an essential trace element in the human body, Mn can be adjusted by metabolism, so it has good biocompatibility.
proposed a simple and convenient method for achieving the controllable growth of ultrathin manganese dioxide nanosheets by introducing a redox reaction on polydopamine nanospheres (Fig. 9). The prepared multifunctional polydopamine@ultrathin manganese dioxide/methylene blue (MB) nanoflowers (termed PDA@ut-MnO2/MB NFs) are suitable platforms for synergistic tumor therapy and can efficiently modulate the tumor microenvironment by generating O2 and depleting GSH. In addition, they can support ultrasensitive reduction-responsive MR imaging (MRI). The morphology and composition of PDA@ut-MnO2 were identified and characterized by scanning electron microscopy (SEM), transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HRTEM) and high-angle annular dark field scanning transmission electron microscopy (HAADF-STEM), and their therapeutic effects were evaluated in mice bearing HCT116 colorectal tumors as models. Tumors were collected 15 days after injection, and PDA@MnO2/MB showed nearly complete tumor growth inhibition under MRI-guided PDT/PTT cotreatment. The H&E staining results of tumor sections also confirmed serious pathological damage, such as severe nuclear pyknosis and nuclear necrosis. Moreover, MRI of tumor-bearing mice found that the T1-weighted MR signal of the kidney was significantly enhanced 2 h after the injection, and the total manganese content in urine was measured by inductively coupled plasma mass spectrometry (ICP-MS). Approximately 65% of the manganese content was detected, suggesting that Mn2+ was eliminated from the body through the kidneys, and the in vivo toxicity was minimal. These results demonstrated that PDA@MnO2/MB NFs can achieve a high degree of tumor growth suppression under MRI-assisted treatment.

 www.ncbi.nlm.nih.gov
www.ncbi.nlm.nih.gov
In this study, natural manganese oxides (MnOx), an environmental material with high redox potential, were used as a promising low-cost oxidant to degrade the widely used dyestuff methylene blue (MB) in aqueous solution. Although the surface area of MnOx was only 7.17 m2 g−1, it performed well in the degradation of MB with a removal percentage of 85.6% at pH 4. It was found that MB was chemically degraded in a low-pH reaction system and the degradation efficiency correlated negatively with the pH value (4–8) and initial concentration of MB (10–50 mg l−1), but positively with the dosage of MnOx (1–5 g l−1). The degradation of MB fitted well with the second-order kinetics. Mathematical models were also built for the correlation of the kinetic constants with the pH value, the initial concentration of MB and the dosage of MnOx. Furthermore, several transformation products of MB were identified with HPLC-MS, which was linked with the bond energy theory to reveal that the degradation was initiated with demethylation.
It has been reported that manganese dioxide possesses high oxidation–reduction potential (1.29 V, 25°C) and that synthetic MnO2 has been applied to degrade phenol and aromatic amines [26,27]. Studies have also found that it performs well in treating bisphenol A, bisphenol AF, bisphenol S and cephalosporins [28,29]. In addition, Fei et al. [30] found that synthesized MnO2 has a high removal efficiency for Congo red. Cheng et al. [31,32] reported that nano-MnO2 could completely destroy HCHO at low temperature. Furthermore, Sekine [33] has improved the performance of nano-MnO2, so that it can be practically used to remove HCHO at room temperature. However, the high cost and complex synthetic steps of synthesized MnO2have limited its application in wastewater treatment. By contrast, manganese ores consisting of multivalent manganese oxides could be a substitute because of abundant reserves in nature. Recently, studies have demonstrated the good degradation efficiency of organic contaminants by natural manganese oxides (MnOx), such as diclofenac [34], melanin [35] and paracetamolin [36] and emerging organic contaminants [37]. MnOx was also extensively used in drinking water treatment for the removal of iron and manganese [38]. However, there are too many different and complex compositions of natural manganese ores. Therefore, it is necessary to find a kind of MnOx with high manganese content and stable effect on removing environmental containments.
It seems mno2 can degrade bpa’s ,reduce glutathione in cancer cells and was combined with methylene blue inhibited tumor growth.
And mno2 was shown to reduce methylene blue and @haidut talked about methylene blue with ascorbic acid and turning it colorless and having some benefits.
This is above my pay grade, is there anything to this?

Strategies to improve photodynamic therapy efficacy by relieving the tumor hypoxia environment - NPG Asia Materials
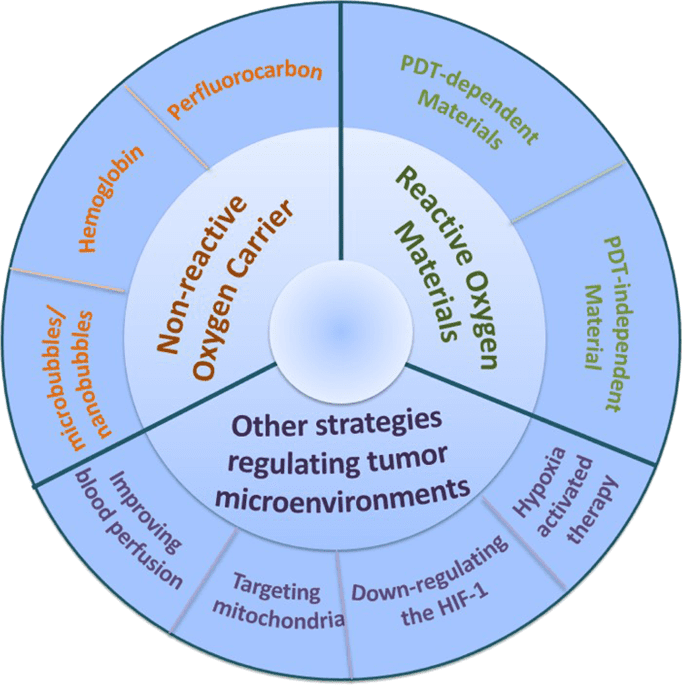
In this review, we summarize the latest progress in the development of strategies to relieve tumor hypoxia for improved PDT efficacy, from the design of novel nonreactive oxygen carriers to reactive materials and other strategies, including the regulation of tumor microenvironments and...
MnO2 nanomaterials.
Manganese oxide (MnO2) has attracted increasing attention due to its ability to generate excessive oxygen with the aid of acid and H2O2 in the tumor microenvironment.The following reactions illustrate how acid-induced H2O2 promotes the degradation of MnO2 and the resultant generation of oxygen:
MnO2 shows high reactivity and specificity to acidic environments and the presence of H2O2, where it can produce oxygen continuously to overcome hypoxia in tumors42,43,44,45. After reacting with excessive H2O2 in an acidic tumor microenvironment, MnO2 nanosheets can be degraded to produce a large amount of oxygen, thus greatly improving the efficiency of PDT. In addition to increasing the oxygen content, MnO2 can also be used as a glutathione (GSH) inhibitor in vivo to increase PDT efficacy. Moreover, it can be used to make upconversion nanocomposites to stimulate PSs at various wavelengths to enhance the selectivity of PDT. In addition, Mn2+ produced by MnO2 is a strong T1 magnetic resonance (MR) contrast agent for tumor detection, which can also be used for in site tumor imaging46,47. At the same time, it is worth mentioning that, as an essential trace element in the human body, Mn can be adjusted by metabolism, so it has good biocompatibility.
MnO2 as a GSH inhibitor to replenish oxygen and increase PDT efficacy
In addition to low oxygen, the effectiveness of PDT is also limited by the hypoxia-induced overexpression of GSH, which consumes ROS before they reach their target and hence greatly reduces the efficiency of PDT. Numerous studies have attempted to develop a more stable and effective PDT system by the incorporation of GSH depletors48. MnO2 can be used as an efficient glutathione dehydrogenase inhibitor to reduce GSH levels; therefore, a MnO2-containing PDT system is expected to overcome the drawbacks caused by hypoxia and GSH overexpression at the tumor site49.proposed a simple and convenient method for achieving the controllable growth of ultrathin manganese dioxide nanosheets by introducing a redox reaction on polydopamine nanospheres (Fig. 9). The prepared multifunctional polydopamine@ultrathin manganese dioxide/methylene blue (MB) nanoflowers (termed PDA@ut-MnO2/MB NFs) are suitable platforms for synergistic tumor therapy and can efficiently modulate the tumor microenvironment by generating O2 and depleting GSH. In addition, they can support ultrasensitive reduction-responsive MR imaging (MRI). The morphology and composition of PDA@ut-MnO2 were identified and characterized by scanning electron microscopy (SEM), transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HRTEM) and high-angle annular dark field scanning transmission electron microscopy (HAADF-STEM), and their therapeutic effects were evaluated in mice bearing HCT116 colorectal tumors as models. Tumors were collected 15 days after injection, and PDA@MnO2/MB showed nearly complete tumor growth inhibition under MRI-guided PDT/PTT cotreatment. The H&E staining results of tumor sections also confirmed serious pathological damage, such as severe nuclear pyknosis and nuclear necrosis. Moreover, MRI of tumor-bearing mice found that the T1-weighted MR signal of the kidney was significantly enhanced 2 h after the injection, and the total manganese content in urine was measured by inductively coupled plasma mass spectrometry (ICP-MS). Approximately 65% of the manganese content was detected, suggesting that Mn2+ was eliminated from the body through the kidneys, and the in vivo toxicity was minimal. These results demonstrated that PDA@MnO2/MB NFs can achieve a high degree of tumor growth suppression under MRI-assisted treatment.

Degradation of methylene blue by natural manganese oxides: kinetics and transformation products
In this study, natural manganese oxides (MnO[x] ), an environmental material with high redox potential, were used as a promising low-cost oxidant to degrade the widely used dyestuff methylene blue (MB) in aqueous solution. Although the surface area of ...
Degradation of methylene blue by natural manganese oxides: kinetics and transformation products
In this study, natural manganese oxides (MnOx), an environmental material with high redox potential, were used as a promising low-cost oxidant to degrade the widely used dyestuff methylene blue (MB) in aqueous solution. Although the surface area of MnOx was only 7.17 m2 g−1, it performed well in the degradation of MB with a removal percentage of 85.6% at pH 4. It was found that MB was chemically degraded in a low-pH reaction system and the degradation efficiency correlated negatively with the pH value (4–8) and initial concentration of MB (10–50 mg l−1), but positively with the dosage of MnOx (1–5 g l−1). The degradation of MB fitted well with the second-order kinetics. Mathematical models were also built for the correlation of the kinetic constants with the pH value, the initial concentration of MB and the dosage of MnOx. Furthermore, several transformation products of MB were identified with HPLC-MS, which was linked with the bond energy theory to reveal that the degradation was initiated with demethylation.
It has been reported that manganese dioxide possesses high oxidation–reduction potential (1.29 V, 25°C) and that synthetic MnO2 has been applied to degrade phenol and aromatic amines [26,27]. Studies have also found that it performs well in treating bisphenol A, bisphenol AF, bisphenol S and cephalosporins [28,29]. In addition, Fei et al. [30] found that synthesized MnO2 has a high removal efficiency for Congo red. Cheng et al. [31,32] reported that nano-MnO2 could completely destroy HCHO at low temperature. Furthermore, Sekine [33] has improved the performance of nano-MnO2, so that it can be practically used to remove HCHO at room temperature. However, the high cost and complex synthetic steps of synthesized MnO2have limited its application in wastewater treatment. By contrast, manganese ores consisting of multivalent manganese oxides could be a substitute because of abundant reserves in nature. Recently, studies have demonstrated the good degradation efficiency of organic contaminants by natural manganese oxides (MnOx), such as diclofenac [34], melanin [35] and paracetamolin [36] and emerging organic contaminants [37]. MnOx was also extensively used in drinking water treatment for the removal of iron and manganese [38]. However, there are too many different and complex compositions of natural manganese ores. Therefore, it is necessary to find a kind of MnOx with high manganese content and stable effect on removing environmental containments.
