Travis
Member
- Joined
- Jul 14, 2016
- Messages
- 3,189
Yeah. Only a few oranges or a pineapple per day will probably prevent the brand of cardiovascular disease that Pauling was talking about; the type dependent on Lp(a) and collagen synthesis. They used megadoses in clinical practice to reverse cardiovascular disease, but Linus Pauling only recommended 2,000 milligrams per day for average people. This is equivalent it 4 L (~1 gallon) of orange juice.* I have actually drank this much before in one day when I worked next to a store that squeezed their own.I found it very useful. I drink a good amount of fresh squeezed OJ every day so I am getting the amount I need.
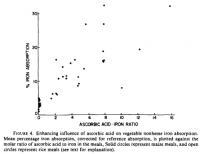
Perhaps I should give more consideration to the iron-absorbing effect of iron. This is very well established, and it's dose-dependent.

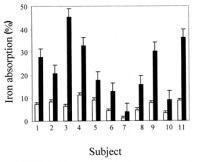
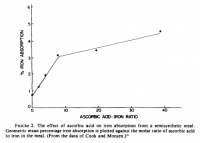 [From 3 separate studies.]
[From 3 separate studies.]But this effect seems to be limited to non-heme iron. Here are some quotes:
Lynch, Sean R., and James D. Cook. "Interaction of vitamin C and iron." Annals of the New York Academy of Sciences 355.1 (1980): 32-44.Similar observations have been made for heme iron, which comes principally from hemoglobin and myoglobin of meat. Heme compounds enter a second common pool and share a set of properties different from those that govern nonheme iron absorption.’ For example, heme is not degraded to any degree in the lumen of the gut but is taken up by the mucosal cells with iron still within the porphyrin ring. Heme iron absorption is relatively independent of other components of the meal; ascorbic acid has no effect.
Turnbull, Adam, Frans Cleton, and Clement A. Finch. "Iron absorption. IV. The absorption of hemoglobin iron." Journal of Clinical Investigation 41.10 (1962): 1897.6. The effect of ascorbic acid on absorption of hemoglobin iron. Ascorbic acid increases the absorption of iron given as ferric salts and of iron in many foods presumably by increasing the amount of ferrous iron presented to the mucosa. Absorption of hemoglobin iron given without food has been found not to be increased by ascorbic acid, and this observation was confirmed in two subjects with iron-deficiency anemia. These subjects, K. A. and B. B., were given 5 mg hemoglobin iron with and without 80 mg ascorbic acid on successive days. Absorptions were 19.3 and 9.6 per cent with ascorbic acid and 14.7 and 9.9 per cent without it, respectively. The effect of a large amount of ascorbic acid on the absorption of hemoglobin iron given with food was studied in three healthy subjects, F. G., D. W., and R. S., who took 2.5 mg hemoglobin iron with a meal, containing 1.8 g iron, with and without 500 mg ascorbic acid on successive days. Absorptions with ascorbic acid were 19.4, 14.3, and 5.6 per cent, averaging 13.1 per cent, and absorptions without it, 27.1, 10.9, and 6.9 per cent, averaging 15.0 per cent. Thus, unlike the absorption of other forms of food iron, that of hemoglobin iron does not appear to be increased by ascorbic acid.
Heme iron is already chelated by the porphyrin ring.The mechanism of action of AA is usually related either to its ability to form soluble iron complexes or its ability to reduce ferric to ferrous ions.
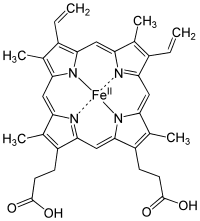
And phytates, vitamin C, and even EDTA have negligible effect on it's absorption.
Likewise, the addition of iron chelators (EDTA) to labeled hemoglobin failed to show a significant decrease in the amount of iron absorbed (15). The absorption of hemoglobin-iron...

Conrad, M. E., et al. "Absorption of hemoglobin iron." American Journal of Physiology--Legacy Content 211.5 (1966): 1123-1130.
Attachments
Last edited: