
Iodine, the secret weapon of hibernating ground squirrels and trauma patients
New work in trauma patients and ground squirrels helps reveal why iodine is so important to life -- and how it could someday be harnessed to improve human health.
www.fredhutch.org
Iodine Redistribution During Trauma, Sepsis, and... : Critical Care Explorations
arctic ground squirrels. Preclinical interventional study using hind-limb ischemia and reperfusion injury in mice. Setting: Level I trauma center emergency room and ICU and animal research laboratories. Subjects: Adult human sepsis and trauma patients, wild-caught adult arctic ground...
The benefits of iodine — an element essential for life — are well established. As a key component of thyroid hormone, it prevents the congenital hypothyroidism that’s the leading preventable cause of intellectual disabilities around the world. Povidone-iodine has also been used in wound care for more than 50 years.
Now, says Dr. Mark Roth, a biochemist at Fred Hutchinson Cancer Research Center, he’s unraveled how iodine interacts with some oxygen-based molecules, reducing cells’ oxygen requirements and protecting against damaging oxygen radicals. The findings, published Oct. 1 in the journal Critical Care Explorations, reveal iodine’s key role in the body’s stress response.
“Iodine is part of a fundamental, primordial, conserved response to inflammation or stress,” explained Roth who studies “metabolic flexibility,” or how organisms from worms to humans can fine-tune their metabolic states.
Metabolically stressful situations, like an injurious car accident or serious bacterial infection, raise cells’ metabolic rates and increase their oxygen consumption. This is accompanied by inflammation and an increase in damaging reactive oxygen species like hydrogen peroxide. Iodine helps on both fronts: It decreases cells’ oxygen consumption and also defangs hydrogen peroxide, turning it into harmless molecules of oxygen and water.
Roth’s work demonstrates that our bodies respond to metabolic stress by detaching iodine atoms from thyroid hormone, freeing it to act as a shield against tissue damage caused by oxygen radicals. A preclinical study in mice showed that giving iodide prior to constricting blood flow to a muscle protects against both local and systemic inflammation-induced tissue damage.
Roth hopes the findings could someday be applied to improve the health of patients facing a tsunami of inflammation — including, perhaps, people experiencing a COVID-19-related cytokine storm. He’s started a company, Faraday Pharmaceuticals, to test iodine’s benefits for patients facing inflammatory tissue damage, such as those undergoing heart attacks or who have undergone trauma. Roth is also collaborating with researchers in the U.S. Army to test whether iodide treatment in the field may improve outcomes for soldiers injured in combat.
Suspended animation leads to an iodized insight
Roth’s work on iodine grew out of in his work on suspended animation, which garnered him a MacArthur Genius Grant. He showed that by using hydrogen sulfide, he could reduce oxygen consumption to the point that small animals, including worms, flies and mice, entered a state of near-metabolic shutdown. But the findings didn’t scale well to larger animals — the difference between an animation-suspending dose of sulfide and a toxic dose was too narrow for safety.So Roth started scanning the periodic table, looking for elements with properties similar to sulfide. These included the elements selenium, bromine, and iodine. Like sulfide, these elements can affect a cell’s (and by extension, an organism’s) metabolic state. The sedating effects of bromide salts are well-known. In fact, in the late 19th and early 20th centuries physicians used them to treat seizure disorders. In some cases, doctors gave patients doses large enough to put them into a coma-like state, dubbed the “bromide sleep.” But Roth was interested in patients’ inflammation instead of their brains.
Iodine was another option. Iodine’s metabolism-calming effects mimic bromine’s, but because iodine’s larger atoms can’t enter cells as easily, it’s less potent. Most famously, iodine is attached to thyroid hormone, which is essential to cellular metabolism. People have understood for thousands of years that ingesting certain foods (now known to be rich in iodine such as seaweed) can prevent an enlarged thyroid gland, or goiter.
But historically, getting enough iodine was difficult. Before salt was iodized, goiter and intellectual disabilities related to iodine deficiency were common in the U.S. (Nowadays, the use of iodine in the dairy and baking industries means that most Americans can get adequate iodine without increasing their salt intake.)
Why should a nutrient so difficult to acquire be necessary?
Iodine’s essentiality has always been attributed to the fact that it’s an integral component of a life-giving molecule, thyroid hormone. But Roth was sure this wasn’t the whole story — it doesn’t describe what iodine itself does. He thinks he’s finally provided the chemical explanation for iodine’s essentiality to life.
Iodine protects against metabolic stress
Roth suspected that iodine’s essentiality may be linked to the way bodies respond to stress. Thyroid hormone gets degraded under metabolically taxing situations, and this frees the attached iodine atoms in a form known as iodide. To see what role iodide could be playing during stressful conditions, Roth and his collaborators looked at what may initially seem like a random collection of sources: hibernating ground squirrels, trauma patients, and patients with sepsis. But there is a connection.“So imagine you're a ground squirrel in the cold, a hibernating ground squirrel,” Roth said. “What you're doing is the same thing you're doing when you're having sepsis. You're trying to lay down and play dead, but not be dead.”
Arctic ground squirrels drop their metabolic rate and reduce their cells’ oxygen consumption every winter, entering a state of low metabolic function called torpor. This state is interspersed with more awake periods, called interbout arousal periods. To measure the iodide levels in the blood of Arctic ground squirrels, Drs. Michael Morrison and Akiko Iwata, the Roth Lab staff scientists who led the study, and research technician Merry Wick, worked with Dr. Kelly Drew at the University of Alaska Fairbanks. They compared blood iodide levels in active summer squirrels and torpid winter squirrels. They found that compared to squirrels during summer or early torpor, the blood iodide content of squirrels in late torpor or interbout arousal doubled or tripled.
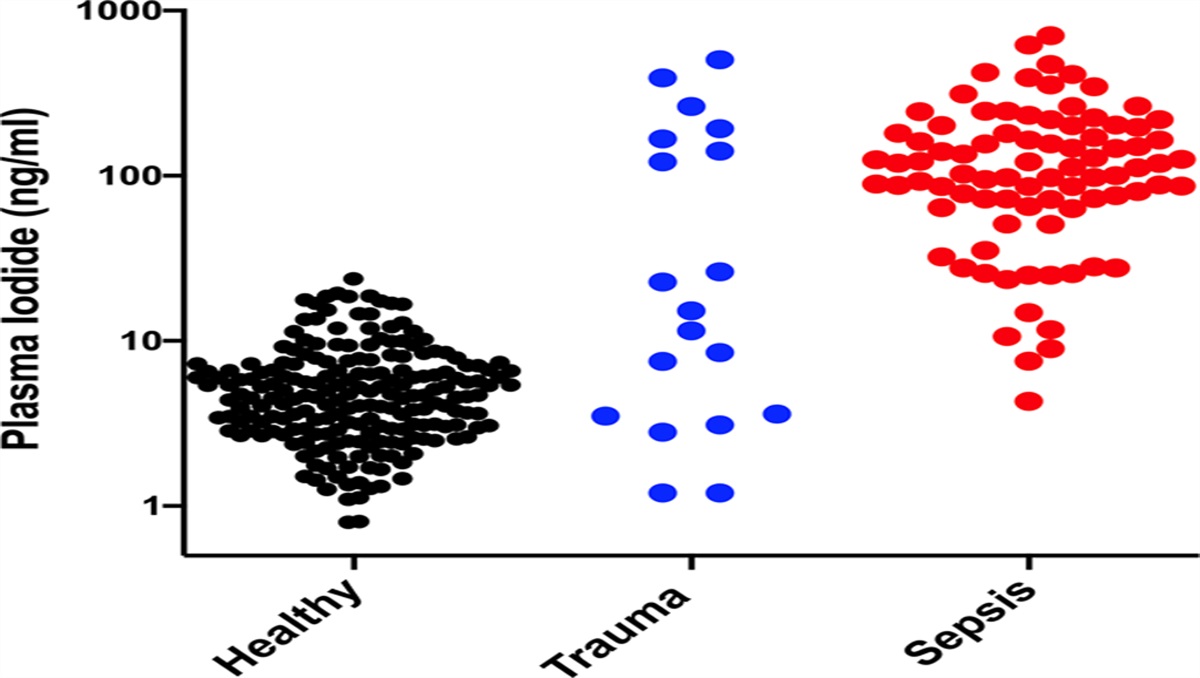
With help from collaborator Dr. Ronald Maier at Harborview Medical Center and the University of Washington, Morrison and Iwata also compared blood iodide levels of patients with sepsis or trauma to healthy donors. They found that in sepsis patients, iodide levels were 26 times higher, and in trauma patients they were 17 times higher, than in healthy people.
This strongly suggested that iodide plays a role in the stress response — but wasn’t a smoking gun. The team then tested the effects of giving extra iodide before a metabolically taxing situation. They found that dosing mice with sodium iodide (one of the compounds used to iodize salt) prior to restricting muscular blood flow reduced inflammation-induced tissue damage.
“We’ve known for many years that stress-induced inflammation makes injuries even worse,” Maier said.
“In this study we found that iodide could provide a recyclable, effective and safe way to block damage from excessive inflammation caused by over production of oxygen radicals after injury and provides a potential therapeutic approach to enhance recovery, prevent complications, and reduce mortality in severely injured patients. The use of iodide in the clinical setting should soon be moving to clinical trials,” he added.
Roth, Morrison and Iwata published in 2018 that giving rats or pigs sodium iodide protected against reperfusion injury in a preclinical model of heart attack. They found that giving pigs sodium iodide prior to inducing a heart attack prevented the usual drop in thyroid hormone that’s associated with increased risk of death.
At the 2019 meeting of the American Heart Association, Faraday announced that a Phase 2 trial showed it’s safe to give patients undergoing heart attacks mega-doses of iodide. The study results support moving to a larger clinical trial to test efficacy, Roth said.
Iodine’s unique chemistry makes it essential
Hydrogen peroxide damages nearby molecules by stealing their electrons. Antioxidants like vitamin C prevent this damage by voluntarily surrendering their electrons to hydrogen peroxide — only to become hungry for electrons themselves.Iodine is different. The individual iodine molecules released from thyroid hormone each carry an extra electron. Known as iodide, this form of iodine sequentially swaps electrons with two molecules of hydrogen peroxide. It ends up where it started: as iodide, ready and able to do it all over again.
“So you get this wonderful agent that can allow you to remain rousable, as it were, in hibernation or [metabolic stress]. And yet it’s still controlling the oxygen consumption associated with inflammation,” Roth said.
He believes this atomic do-si-do explains why iodine atoms are linked to thyroid hormone. Because thyroid hormones increase oxygen consumption, they also increase inflammation and oxidative damage. Iodine’s there to balance it out. It’s also why kelp needs iodine, he said — to shield against oxidative stress caused by the UV rays that scorch it at low tide. It’s the echo of a primordial chemical dance that began in our planet’s infancy, long before the first stirrings of life.
From basic chemistry to human health
The jump in iodide blood levels that Morrison and Roth saw in trauma and sepsis patients is enormous, suggesting a powerful biochemical response is occurring, Roth said. (In contrast, lactate levels only rise about three- to four-fold in patients experiencing organ damage.) But it will take several steps to go from the insight that iodine plays a central role in alleviating metabolic stress to improving human health.The findings open up several avenues for further exploration. This includes determining whether a small daily dose of iodide, or a one-time megadose, provides more benefits. Roth is working with the U.S. Army to address this question.
Patients with COVID-19 are an obvious group who might benefit from an inflammation-dampening treatment — and Roth’s also helping to test that idea. With Maier, Roth is planning to test iodine levels from COVID patients to see if their levels increase similarly to trauma and sepsis patients (as he expects they will). Roth is also working with collaborators at the National Institute of Allergy and Infectious Diseases to test whether sodium iodide administration affects outcomes of animals infected with the novel coronavirus.
These strategies seek to capitalize on the normal ebb and flow between hydrogen peroxide and iodine. As inflammation edges upward, iodine reins it in — and a megadose of iodine may be needed when inflammation outpaces our bodies’ own stores.
“What you're really trying to do is oscillate. That is to say, your heartbeat, your breathing — you're sleeping, you're awake,” he said. “And when you get right down to why these things were chosen [for life], it's the fundamental oscillators of life.”
Note: Scientists at Fred Hutch played a role in developing these discoveries, and Fred Hutch and certain of its scientists may benefit financially from this work in the future.
This work was funded by the Army Research Office and the National Science Foundation.
Last edited:


