TreasureVibe
Member
- Joined
- Jul 3, 2016
- Messages
- 1,941
Hey all, I'm asking @haidut @burtlancast @Travis and the rest of you to review the following thesis:
Cancer is a disease, in which a cell goes into a state of fermentation, causing it to become a cancer cell. This explains the Warburg effect seen by Dr. Warburg with lactic acid. Cancer = cell fermentation. See the following:
Fermentation and anaerobic respiration
How cells extract energy from glucose without oxygen. In yeast, the anaerobic reactions make alcohol, while in your muscles, they make lactic acid.
2015 study:
How Fermentation Gives Us Beer, Wine, Cheese—and Cancer?
Even in the presence of oxygen, cancer cells and some bacteria prefer fermentation, a new study finds
By Bret Stetka on December 15, 2015
Wine, beer and yogurt are produced when microorganisms convert sugar into alcohol, gases or acids. But this process of fermentation—which is used by bacteria, fungi and other fast-growing cells to generate energy in the absence of oxygen—is a much less efficient way of generating energy for cells than aerobic respiration. Credit: ©iStock
In 1931 German physician, physiologist and biochemist Otto Heinrich Warburg won the Nobel Prize for his discovery that cancerous cells—unlike most healthy human cells, which produce energy using oxygen via respiration—favor the anaerobic process of fermentation, or the conversion of sugar into acids, gases or alcohol, even in the presence of oxygen. This has perplexed scientists ever since because fermentation is a far less efficient means of generating energy than aerobic metabolism, hence its pejorative tag as a “wasteful metabolism.”
Gaston Naessens said there were micro organisms within cells, that can transform into certain bacteria or fungi. He called these micro organisms somatids. These micro organisms can start the state of fermentation. This fermentation causes the cell to become a cancer cell. This explains why there are reports of all types of fungi and bacteria being found inside tumors. These are not the actual fungi or bacteria, but the somatids transformed into one. These somatids could be the body's own pseudo-pathogens/pseudo-bacteria/pseudo-fungi. Entities that can transform into any pathogen of their liking. Perhaps to keep energy metabolism of the cell going in the absence of oxygen or another reason. The cell switches to using fermentation for energy production, in which the cell becomes a cancer cell, or a pre-cancer cell. Gaston Naessens cured cancer by giving these micro organisms nitrogen. This stopped them from excreting toxins, and either made the immune system succesfully kill the cell, or reversed the cancer cell into a healthy cell again. Nitrogen is essential in Yeast Assimilable Nitrogen (YAN) to fully go through successful fermentation.
See following sources too.
I had the feeling I was reading about the human body when I read this Wikipedia article on yeast assimilable nitrogen: Yeast assimilable nitrogen - Wikipedia anti-cancer nutrients/substances which are popular on the forum here like urea and glycine are actually implied in yeast assimilable nitrogen, which is a substance used in wine fermentation/wine making. You can find those nutrients/substances back in the Wikipedia article on yeast assimilable nitrogen.
The Somatid, Gaston Naessens, 714X Cancer Treatment (last post especially)
Ted from Bangkok said
"Here is how the cancer switch is turned on: If I am a weak person, whose healthy cells are around -60 millivolts, and I take extra long shower with chlorinated water, whose millivolts is -300 and our body ABSORBS an average of 80% of the chlorine through our skin as chlorine is a gas. What you are doing in effect is to change your ALREADY low millivolts -60 into exactly -15 millivolts and suddenly the electrical energy now low, the mitochondrial now goes into anaerobic fermentation and becomes cancerous. This can happen if one were to be consuming an acid form of vitamin C, and other acid drinks. The only way to prove that is vitamin C in acid form is actually positive millivolts, usually 150 to 300, depending on various things.
Here is how the cancer switch is turned on: If I am a weak person, whose healthy cells are around -60 millivolts, and for instance I take an extra long shower with chlorinated water, whose millivolts are -300, your body ABSORBS an average of 80% of the chlorine through your skin as chlorine gas. What you are doing in effect is to change your ALREADY low millivolts -60 into exactly -15 millivolts and suddenly the mitochondria go into anaerobic fermentation and those cells become cancerous."
Dr Pantellini would change the voltage of the cell back to a healthy one using potassium, reverting the state of fermentation perhaps, or ''killing'' the pathogen inside, which reverted the state of fermentation, or got the cell killed by the immune system. Dr. Revici used lipidic potassium to kill the pathogen inside which would revert the state of fermentation or got the cell killed by the immune system. Gaston Naessens fed the cell nitrogen (known in fermentation in wine) and either reverted the state of fermentation or got the cell killed by the immune system
It's time to connect the dots, folks!
Cancer = cell fermentation. The cell being in a state of fermentation.
Why it goes into a state of fermentation could have multiple reasons, one being compromising for energy loss in the absence of oxygen, a glitch in the mitochondria or cell, a pathogen hijacking the cell from the somatid, any reason. It can even go into a state of fermentation if there is an oxygen supply available, the study above demonstrates.
But the fact that there is a ''state of fermentation'' for a cell is something that is not yet recognized broadly, but is probably what defines a cancer cell, or a pre-stage of it. The 2015 study above supports that.
The micro-organism which can mimick any pathogen of its liking which resides within the cell found by Naessens is the missing link.
It could be a theory that any pathogen displaces or replaces the somatid, hijacking the cell, and starting a state of fermentation, causing the cell to become cancerous. Therefore implying a viral/bacterial/fungal implication for the disease of cancer too. Electricity, voltage, like Ted mentions above, could also be a reason why the cell decides to go into a state of fermentation.
Perhaps the state of fermentation is to avoid necrosis?
Necrosis - Wikipedia
Necrosis - Wikipedia
Cancer is a disease, in which a cell goes into a state of fermentation, causing it to become a cancer cell. This explains the Warburg effect seen by Dr. Warburg with lactic acid. Cancer = cell fermentation. See the following:
Fermentation and anaerobic respiration
How cells extract energy from glucose without oxygen. In yeast, the anaerobic reactions make alcohol, while in your muscles, they make lactic acid.
Source: Fermentation and anaerobic respirationIntroduction
Ever wonder how yeast ferment barley malt into beer? Or how your muscles keep working when you're exercising so hard that they're very low on oxygen?
Both of these processes can happen thanks to alternative glucose breakdown pathways that occur when normal, oxygen-using (aerobic) cellular respiration is not possible—that is, when oxygen isn't around to act as an acceptor at the end of the electron transport chain. These fermentation pathways consist of glycolysis with some extra reactions tacked on at the end. In yeast, the extra reactions make alcohol, while in your muscles, they make lactic acid.
Fermentation is a widespread pathway, but it is not the only way to get energy from fuels anaerobically (in the absence of oxygen). Some living systems instead use an inorganic molecule other than \text {O}_2O2O, start subscript, 2, end subscript, such as sulfate, as a final electron acceptor for an electron transport chain. This process, called anaerobic cellular respiration, is performed by some bacteria and archaea.
In this article, we'll take a closer look at anaerobic cellular respiration and at the different types of fermentation.
Anaerobic cellular respiration
Anaerobic cellular respiration is similar to aerobic cellular respiration in that electrons extracted from a fuel molecule are passed through an electron transport chain, driving \text{ATP}ATPA, T, P synthesis. Some organisms use sulfate (\text {SO}_4^{2-})(SO42−)left parenthesis, S, O, start subscript, 4, end subscript, start superscript, 2, minus, end superscript, right parenthesis as the final electron acceptor at the end ot the transport chain, while others use nitrate (\text {NO}_{3}^-)(NO3−)left parenthesis, N, O, start subscript, 3, end subscript, start superscript, minus, end superscript, right parenthesis, sulfur, or one of a variety of other molecules^11start superscript, 1, end superscript.
What kinds of organisms use anaerobic cellular respiration? Some prokaryotes—bacteria and archaea—that live in low-oxygen environments rely on anaerobic respiration to break down fuels. For example, some archaea called methanogens can use carbon dioxide as a terminal electron acceptor, producing methane as a by-product. Methanogens are found in soil and in the digestive systems of ruminants, a group of animals including cows and sheep.
Similarly, sulfate-reducing bacteria and Archaea use sulfate as a terminal electron acceptor, producing hydrogen sulfide (\text H_2\text S)(H2S)left parenthesis, H, start subscript, 2, end subscript, S, right parenthesis as a byproduct. The image below is an aerial photograph of coastal waters, and the green patches indicate an overgrowth of sulfate-reducing bacteria.

Aerial photograph of coastal waters with blooms of sulfate-reducing bacteria appearing as large patches of green in the water.
Image credit: "Metabolism without oxygen: Figure 1," OpenStax College, Biology, CC BY 3.0; Modification of work by NASA/Jeff Schmaltz, MODIS Land Rapid Response Team at NASA GSFC, Visible Earth Catalog of NASA images.
Fermentation
Fermentation is another anaerobic (non-oxygen-requiring) pathway for breaking down glucose, one that's performed by many types of organisms and cells. In fermentation, the only energy extraction pathway is glycolysis, with one or two extra reactions tacked on at the end.
Fermentation and cellular respiration begin the same way, with glycolysis. In fermentation, however, the pyruvate made in glycolysis does not continue through oxidation and the citric acid cycle, and the electron transport chain does not run. Because the electron transport chain isn't functional, the \text{NADH}NADHN, A, D, Hmade in glycolysis cannot drop its electrons off there to turn back into \text {NAD}^+NAD+N, A, D, start superscript, plus, end superscript
The purpose of the extra reactions in fermentation, then, is to regenerate the electron carrier \text{NAD}^+NAD+N, A, D, start superscript, plus, end superscript from the \text{NADH}NADHN, A, D, H produced in glycolysis. The extra reactions accomplish this by letting \text{NADH}NADHN, A, D, H drop its electrons off with an organic molecule (such as pyruvate, the end product of glycolysis). This drop-off allows glycolysis to keep running by ensuring a steady supply of \text{NAD}^+NAD+N, A, D, start superscript, plus, end superscript.
Lactic acid fermentation
In lactic acid fermentation, \text{NADH}NADHN, A, D, H transfers its electrons directly to pyruvate, generating lactate as a byproduct. Lactate, which is just the deprotonated form of lactic acid, gives the process its name. The bacteria that make yogurt carry out lactic acid fermentation, as do the red blood cells in your body, which don’t have mitochondria and thus can’t perform cellular respiration.
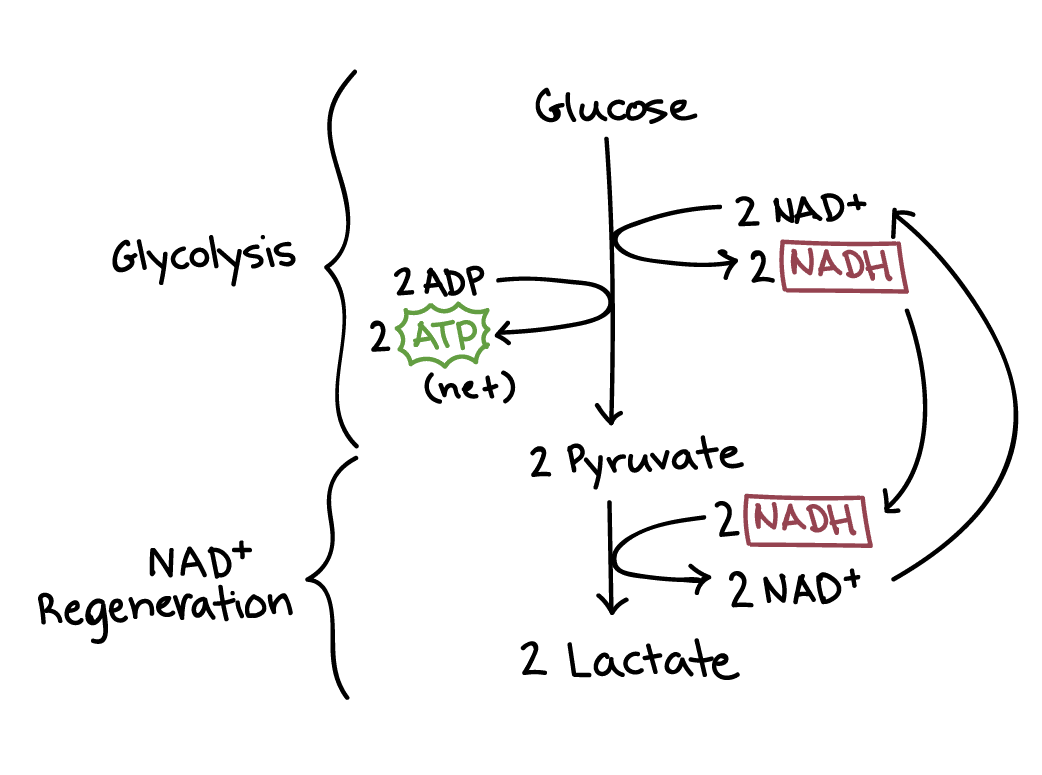
Diagram of lactic acid fermentation. Lactic acid fermentation has two steps: glycolysis and NADH regeneration.
During glycolysis, one glucose molecule is converted to two pyruvate molecules, producing two net ATP and two NADH.
During NADH regeneration, the two NADH donate electrons and hydrogen atoms to the two pyruvate molecules, producing two lactate molecules and regenerating NAD+.
Muscle cells also carry out lactic acid fermentation, though only when they have too little oxygen for aerobic respiration to continue—for instance, when you’ve been exercising very hard. It was once thought that the accumulation of lactate in muscles was responsible for soreness caused by exercise, but recent research suggests this is probably not the case.
Lactic acid produced in muscle cells is transported through the bloodstream to the liver, where it’s converted back to pyruvate and processed normally in the remaining reactions of cellular respiration.
Alcohol fermentation
Another familiar fermentation process is alcohol fermentation, in which \text{NADH}NADHN, A, D, Hdonates its electrons to a derivative of pyruvate, producing ethanol.
Going from pyruvate to ethanol is a two-step process. In the first step, a carboxyl group is removed from pyruvate and released in as carbon dioxide, producing a two-carbon molecule called acetaldehyde. In the second step, \text{NADH}NADHN, A, D, H passes its electrons to acetaldehyde, regenerating \text{NAD}^+NAD+N, A, D, start superscript, plus, end superscript and forming ethanol.
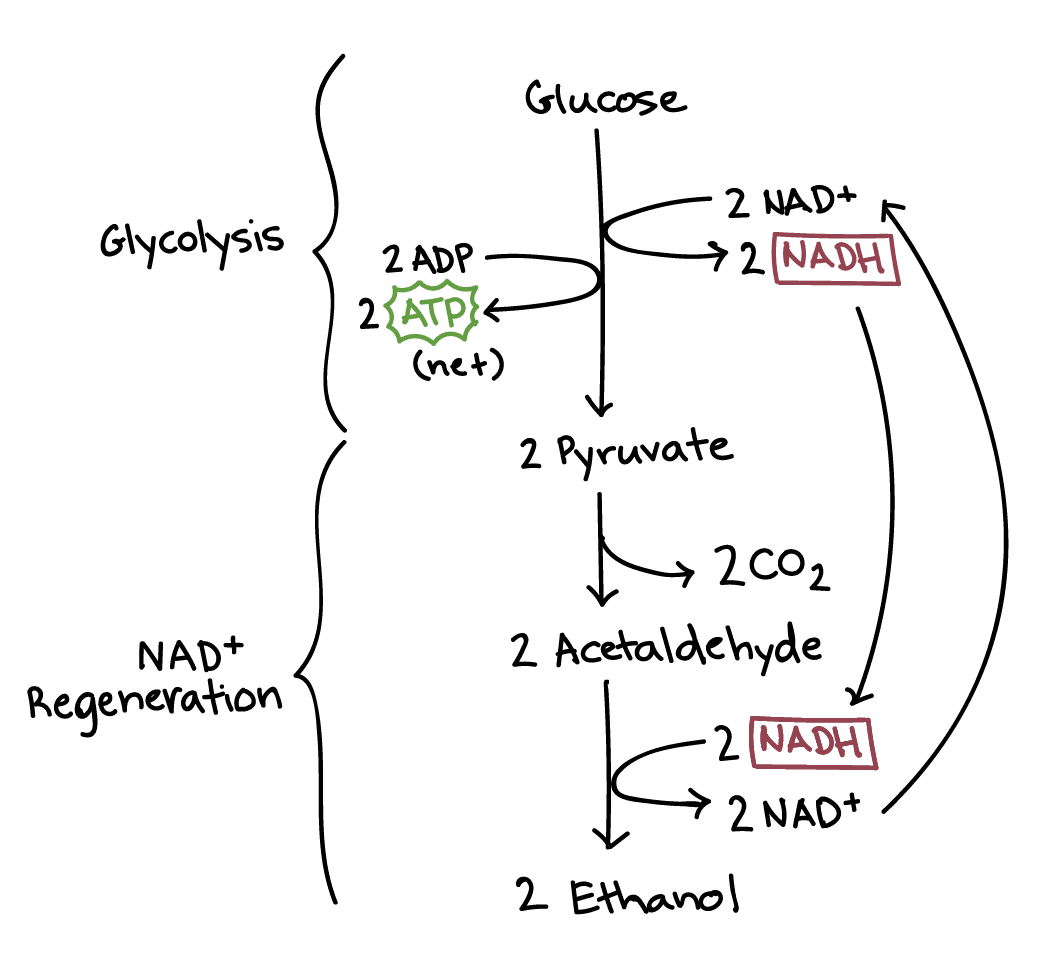
Diagram of alcohol fermentation. Alcohol fermentation has two steps: glycolysis and NADH regeneration.
During glycolysis, one glucose molecule is converted to two pyruvate molecules, producing two net ATP and two NADH.
During NADH regeneration, the two pyruvate molecules are first converted to two acetaldehyde molecules, releasing two carbon dioxide molecules in the process. The two NADH then donate electrons and hydrogen atoms to the two pyruvate molecules, producing two ethanol molecules and regenerating NAD+.
Alcohol fermentation by yeast produces the ethanol found in alcoholic drinks like beer and wine. However, alcohol is toxic to yeasts in large quantities (just as it is to humans), which puts an upper limit on the percentage alcohol in these drinks. Ethanol tolerance of yeast ranges from about 555 percent to 212121 percent, depending on the yeast strain and environmental conditions.
Facultative and obligate anaerobes
Many bacteria and archaea are facultative anaerobes, meaning they can switch between aerobic respiration and anaerobic pathways (fermentation or anaerobic respiration) depending on the availability of oxygen. This approach allows lets them get more ATP out of their glucose molecules when oxygen is around—since aerobic cellular respiration makes more ATP than anaerobic pathways—but to keep metabolizing and stay alive when oxygen is scarce.
Other bacteria and archaea are obligate anaerobes, meaning they can live and grow only in the absence of oxygen. Oxygen is toxic to these microorganisms and injures or kills them on exposure. For instance, the Clostridium bacteria that are responsible for botulism (a form of food poisoning) are obligate anaerobes^22start superscript, 2, end superscript. Recently, some multicellular animals have even been discovered in deep-sea sediments that are free of oxygen^{3,4}3,4start superscript, 3, comma, 4, end superscript.
Self-check

Image of tanks used for wine production by fermentation of grapes. The tanks are quipped with pressure-release valves.
Image credit: "Metabolism without oxygen: Figure 3" by OpenStax College, Biology, CC BY 3.0
- Inside these tanks, yeasts are busily fermenting grape juice into wine. Why do winemaking tanks like these need pressure-release valves?
Choose 1 answer:
Choose 1 answer:
- (Choice A)
A
The yeasts produce \text O_2O2O, start subscript, 2, end subscript gas by cellular respiration.- (Choice B)
B
The yeasts produce \text {CO}_2CO2C, O, start subscript, 2, end subscript gas by lactic acid fermentation.- (Choice C)
C
The yeasts produce \text {CO}_2CO2C, O, start subscript, 2, end subscript gas by alcohol fermentation.- (Choice D)
D
The yeasts produce \text {CO}_2CO2C, O, start subscript, 2, end subscript gas by cellular respiration.
2015 study:
How Fermentation Gives Us Beer, Wine, Cheese—and Cancer?
Even in the presence of oxygen, cancer cells and some bacteria prefer fermentation, a new study finds
By Bret Stetka on December 15, 2015
Wine, beer and yogurt are produced when microorganisms convert sugar into alcohol, gases or acids. But this process of fermentation—which is used by bacteria, fungi and other fast-growing cells to generate energy in the absence of oxygen—is a much less efficient way of generating energy for cells than aerobic respiration. Credit: ©iStock
In 1931 German physician, physiologist and biochemist Otto Heinrich Warburg won the Nobel Prize for his discovery that cancerous cells—unlike most healthy human cells, which produce energy using oxygen via respiration—favor the anaerobic process of fermentation, or the conversion of sugar into acids, gases or alcohol, even in the presence of oxygen. This has perplexed scientists ever since because fermentation is a far less efficient means of generating energy than aerobic metabolism, hence its pejorative tag as a “wasteful metabolism.”
But a team of scientists from the University of California, San Diego, has discovered that although oxygen-based metabolism is a more efficient means of energy production, the costs required to produce the molecular machinery that drives respiration are twice those needed to ferment the sugar glucose. Their work could have implications in identifying potential targets in treating cancer.
The team measured what is called proteome allocation—or the fraction of all cellular proteins devoted to various tasks—to determine the metabolic costs of generating energy and cell growth in Escherichia coli bacteria. The enzymes that facilitate respiration—the raw machinery that normally supports human cellular life—are large and lumbering and need to be produced prolifically to keep us, and our steadily growing cells, going. Put another way, a higher percentage of a fast-growing cell’s proteome is dedicated to growth whereas a smaller fraction is available for other cellular processes, including energy production.
University of California, San Diego, physics and biology professor Terry Hwa, who led the study, likens his findings, recently published in Nature,to coal versus nuclear energy. "Coal factories produce energy less efficiently than nuclear power plants on a per-carbon basis, but they are a lot cheaper to build,” he said in a statement. So the decision of which route to generate energy depends on the availability of coal and the available budget for building power plants.” Fast-growing cells find fermentation the cheaper path. In this sense it is coal energy for cells. (Scientific American is part of Nature Publishing Group.)
The idea that cellular metabolism and growth might be based on the cost-benefit balance of producing the proteins necessary to generate energy and grow was first proposed by a team of Dutch theoretical biologists in 2009. Hwa’s findings confirm those findings. And although prevailing dogma views cancer as a genetic disorder—or really a complex of disorders caused by countless possible mutations—some researchers are coming around to the idea that the ultimate pathologic insult might be impaired or altered energy production.
Thomas Seyfried, a biologist at Boston College who was not part of this study, feels that cancer is a metabolic disorder, citing the large body of evidence implicating mitochondrial dysfunction in cancer. Mitochondria—or the “powerhouses” of our cells—are where cellular energy production takes place. “There is now substantial evidence from a broad range of disciplines showing some degree of defect in the number, structure or function of mitochondria in all types of tumor cells. These mitochondrial defects cause the enhanced glucose uptake and the fermentation seen in tumor cells,” Seyfried explains.
In a 2014 paper by Seyfried and colleagues published in Carcinogenesis he cites ample evidence to support his claim, including showing that a cell’s tumor potential is suppressed if it is transplanted with normal mitochondria; and conversely that transferring mitochondria from tumor cells into the cytoplasm of normal cells increases the chances that those once normal cells will become cancerous. He also points out the large body of work connecting the etiological dots: Many of the mutated genes associated with cancer seem to exert their effects by impairing cellular respiration. It is also possible, Seyfried strongly feels, that transitioning from respiration to fermentation produces free radicals that cause genetic mutations associated with cancer.
Seyfried also suggests a possible evolutionary explanation for fermentation in cancer cells, citing work by Carlos Sonnenschein and Ana Soto at Tufts University showing that the default state for cells is to proliferate, like cancer cells do, and that aerobic respiration in the mitochondria normally helps keep this growth in check. “Unbridled proliferation driven by fermentation metabolism was the state of existence for most cells before oxygen entered the atmosphere some two billion years ago,” he explains. “A gradual loss of respiratory control together with a compensatory fermentation underlies the origin of cancer.”
The association between energy production and cancer is likely far from being completely understood, and although Hwa cautions that he is not a cancer biologist, he feels there is definite promise in pursuing treatments that tinker with metabolism. “I can see that interfering with fermentation could be an effective strategy to slow down tumor growth,” he explains, “since slow-growing cells rely more on respiration to generate energy—then, in principle, this treatment strategy is naturally more disruptive to fast-growing cancer cells than normal cells.”
Current cancer treatment emphasizes interfering with cell signaling pathways that could lead to runaway cellular growth. “But from this study,” Hwa says, “[we found] that maybe we don't need to be so concerned with signaling and could instead work to slow down the efficiency of fermentative processes. We can then count on cancer cells’ growth to slow down as they shift to respiration.”
As more and more mutations associated with varying cancers are uncovered, developing oncology therapies could seem a Sisyphean undertaking. But a single pathology—one that perhaps results in the mutations associated with cancer—could make developing effective cancer therapies a whole lot easier.
As Otto Warburg’s work alluded to nearly a century ago, perhaps this entails simply encouraging cancer to take a breath of fresh air.
Gaston Naessens said there were micro organisms within cells, that can transform into certain bacteria or fungi. He called these micro organisms somatids. These micro organisms can start the state of fermentation. This fermentation causes the cell to become a cancer cell. This explains why there are reports of all types of fungi and bacteria being found inside tumors. These are not the actual fungi or bacteria, but the somatids transformed into one. These somatids could be the body's own pseudo-pathogens/pseudo-bacteria/pseudo-fungi. Entities that can transform into any pathogen of their liking. Perhaps to keep energy metabolism of the cell going in the absence of oxygen or another reason. The cell switches to using fermentation for energy production, in which the cell becomes a cancer cell, or a pre-cancer cell. Gaston Naessens cured cancer by giving these micro organisms nitrogen. This stopped them from excreting toxins, and either made the immune system succesfully kill the cell, or reversed the cancer cell into a healthy cell again. Nitrogen is essential in Yeast Assimilable Nitrogen (YAN) to fully go through successful fermentation.
See following sources too.
I had the feeling I was reading about the human body when I read this Wikipedia article on yeast assimilable nitrogen: Yeast assimilable nitrogen - Wikipedia anti-cancer nutrients/substances which are popular on the forum here like urea and glycine are actually implied in yeast assimilable nitrogen, which is a substance used in wine fermentation/wine making. You can find those nutrients/substances back in the Wikipedia article on yeast assimilable nitrogen.
The Somatid, Gaston Naessens, 714X Cancer Treatment (last post especially)
Ted from Bangkok said
"Here is how the cancer switch is turned on: If I am a weak person, whose healthy cells are around -60 millivolts, and I take extra long shower with chlorinated water, whose millivolts is -300 and our body ABSORBS an average of 80% of the chlorine through our skin as chlorine is a gas. What you are doing in effect is to change your ALREADY low millivolts -60 into exactly -15 millivolts and suddenly the electrical energy now low, the mitochondrial now goes into anaerobic fermentation and becomes cancerous. This can happen if one were to be consuming an acid form of vitamin C, and other acid drinks. The only way to prove that is vitamin C in acid form is actually positive millivolts, usually 150 to 300, depending on various things.
Here is how the cancer switch is turned on: If I am a weak person, whose healthy cells are around -60 millivolts, and for instance I take an extra long shower with chlorinated water, whose millivolts are -300, your body ABSORBS an average of 80% of the chlorine through your skin as chlorine gas. What you are doing in effect is to change your ALREADY low millivolts -60 into exactly -15 millivolts and suddenly the mitochondria go into anaerobic fermentation and those cells become cancerous."
Dr Pantellini would change the voltage of the cell back to a healthy one using potassium, reverting the state of fermentation perhaps, or ''killing'' the pathogen inside, which reverted the state of fermentation, or got the cell killed by the immune system. Dr. Revici used lipidic potassium to kill the pathogen inside which would revert the state of fermentation or got the cell killed by the immune system. Gaston Naessens fed the cell nitrogen (known in fermentation in wine) and either reverted the state of fermentation or got the cell killed by the immune system
It's time to connect the dots, folks!
Cancer = cell fermentation. The cell being in a state of fermentation.
Why it goes into a state of fermentation could have multiple reasons, one being compromising for energy loss in the absence of oxygen, a glitch in the mitochondria or cell, a pathogen hijacking the cell from the somatid, any reason. It can even go into a state of fermentation if there is an oxygen supply available, the study above demonstrates.
But the fact that there is a ''state of fermentation'' for a cell is something that is not yet recognized broadly, but is probably what defines a cancer cell, or a pre-stage of it. The 2015 study above supports that.
The micro-organism which can mimick any pathogen of its liking which resides within the cell found by Naessens is the missing link.
It could be a theory that any pathogen displaces or replaces the somatid, hijacking the cell, and starting a state of fermentation, causing the cell to become cancerous. Therefore implying a viral/bacterial/fungal implication for the disease of cancer too. Electricity, voltage, like Ted mentions above, could also be a reason why the cell decides to go into a state of fermentation.
Perhaps the state of fermentation is to avoid necrosis?
Necrosis - Wikipedia
Necrosis - Wikipedia
Last edited:
